Trypanosoma cruzi: pathogenesis, epidemiology, and recent developments in the potential treatment of Chagas' disease
Background and Significance

By Sriya Chadalavada
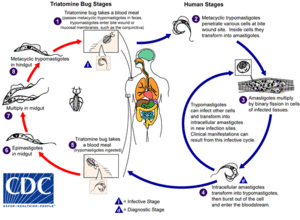
Trypanosoma cruzi is a protozoan parasite that is most frequently transmitted through triatomine bugs. The genome of T. cruzi was fully sequenced in 2005. [8] Triatomine bugs belong to the Rediviidae family and are colloquially known as “kissing bugs” due to sucking blood near the victim’s face when feeding. Another common name they are known by is "assassin bugs." [4] The infection of T. cruzi results in a lifelong condition known as American trypanosomiasis or Chagas disease. Although only a few species are known to cause Chagas disease, over 130 Triatomine bugs species have been identified. [6] At least eleven species of triatomine bugs are confirmed to be capable of transmitting the T. cruzi parasite. [2] Of these Triatoma infestans, Rhodnius prolixus, and Triatoma dimidiate result in the highest number of infections. Triatonimes themselves appear to be unaffected by T. cruzi infections. [6]
There is less research and international concern for T. cruzi and Chagas disease than what many scientists believe is required. The World Health Organization (WHO) has categorized Chagas Disease as “one of the world’s 13 most neglected tropical diseases.” [6] Although the disease and parasite were discovered over a century ago, there remains to be a lack in scientific efforts for better treatment and cures. The human infection of Chagas disease is endemic to Latin America. [11] Although Chagas disease was once centered in Latin America, it has gradually spread across the world (Figure 1).[6] Chagas, a Brazillian physician initially identified the disease in 1909. Later, it was discovered that Chagas disease had afflicted humans at least as early as 9000 years ago when T. cruzi DNA was isolated from human mummies. Some historians believe that Charles Darwin may have suffered from Chagas Disease according to his recorded accounts of coming in contact with triatomine in South America and symptoms later in life. [6] Since the turn of the century, the incidence rates of Chagas disease has greatly decreased. [11]
Humans are typically bitten when asleep. As the face is often left uncovered, most bites occur in this area. Prior to feeding, kissing bugs extend mouthparts from underneath their body. It is then used to feed on blood. [12] Feeding can continue for up to a few minutes. If interrupted when feeding, kissing bugs may leave a cluster of bites. Defecating during feeding is common. [4] Chagas disease spreads to humans when the fecal matter of T. cruzi enters the bloodstream through a break in the skin. Very rarely, it can also be transmitted if consumed with food or drink. [2][6]
T. cruzi is a hemoflagellate, meaning it can live in the bloodstream and has flagella. T. cruzi has a life cycle with three stages: epimastigotes, trypomastigotes, and amastigotes (Figure 2). Replication occurs at the epimastigotic stage, typically in the midgut of kissing bugs. They then become trypomastigotes. This process differentiates the stage from when T. cruzi is nonpathogenic to becoming pathogenic. They are non-replicative. When the insect defecates, the parasite is also released and has the ability to enter the bloodstream and infect a mammalian host. Once inside cells, the T. cruzi begin transforming to amastigotes, the principal form of the parasite in vertebrate hosts. [4] T. cruzi replicate through binary fission. [13]While other mammalian tryposomes divide in the bloodstream, T. cruzi can only replicate in cells. [21] Following replication for multiple generations, they then become trypomastigotes before leaving infected cells for new cells. They then alternate between these last two stages as they enter and leave cells. [14]
There has been research on additional physiological, biochemical, and pathological variations between these forms. As epimastigotes transform to trypomastigotes, there are notable changes to multiple cellular components, primarily within the kinetoplast DNA network and with lipid inclusions. Soares et al. studied whether there were any changes to the reservosome during this metamorphosis. A reservosome is where proteins ingested by the parasite and lipids would collect. The reservosome is known to occupy around 5.6% of epimastigotes cell volume. It was found that during this stage of differentiation from an epimastigote to a trypomastigote, the parasite accumulates significantly more lipids in the reservosome to use as its primary energy reserve. Thus, Soares et al., showed that there were changes to the lipid composition of the reservosome during this stage. [17] Epimastigotes exhibit lower negative surface charges than trypomastigotes. This was found to be due to the presence of increased sialic acid on the surface of the trypomastigotic form. [19] The epimastigotic form of T. cruzi fixes CO2 as facultative anaerobe and produces succinate during glucose metabolism. It is believed that either PEP carboxykinase or malic enzyme is responsible for CO2 fixation. [23]
Epidemiology
Reduviidae can be found across the world, but Tritominae are specific to the Americas. [12] They prefer to live in grassy or wooded regions. Triatome bugs are attracted to and gather near black lights. [4] [10] They also have a strong preference for warm climates. [12] June is the month when the most bites are reported. [4] Factors such as deforestation and habitat loss may contribute to the spread and growing prevalence of triatomines, particularly in rural regions of Latin America. 11] The North American population of Triatome bugs is continuously expanding. Although additional research is necessary to make this claim, some believe that global warming is contributing the spread of these bugs northwards. [4]
In some regions studied, half of the triatomine bugs contained the parasite. [2] In one survey, 25% of the bugs collected in the Tuscan, Arizona area contained T. cruzi and this ratio was almost 50% in San Diego, California. They obtain T. cruzi when feeding on mammalian hosts of this parasite. The more blood triatonimes have consumed, the higher the probability that they carry the parasite; thus, it is the oldest triatonimes that are most likely to be infections. [6] The following animals have been found particularly vulnerable to the parasite: rodents, possums, raccoons, armadillos, skunks, and dogs, cats, chickens. [2] [3] In the United States, there are approximately 300,000 people living with Chagas disease, most of which are Latin American immigrants. [2] [4] Only six cases of direct bug to human transmission have been reported in the United States. Concurrently, in Latin America there are around 8 million people living with Chagas disease. The disparity in infections between these regions is notable.
This disparity is thought to be in part due to the unsuitability of Triatonite bug colonization in homes in the United States. [4]. This is a socioeconomic factor of infestation. In homes built of mud, sticks, and tiles, there is a greater chance of kissing bug infestation. Such homes are more common in Latin America. In homes built of cement and brick, a housing style customary in the United States, there was a greatly lowered chance of infestation. Another reason could be the absence of certain foliage in proximity to homes in the United States. One study found that kissing bugs were more prevalent in Latin American homes with certain fruit-bearing trees such as avocado or coffee. This was thought to be due to the specific leaf litter from the trees enabling an ideal breeding location. [3] Other factors could include the bugs preferring other mammalian hosts over humans and delayed defecation after biting [4].
Another method of control is the use of synthetic parathyroid insecticides. Depending on the surface used, the insecticide may be effective from 2 months up to a year. Resistance among kissing bugs is very low since they have long lifespans and low tendencies of genetic variability. [5] In South and Central America, the main form of transmission to humans is from insects. Chagas disease is more prevalent in the poor and rural areas of this region. In the 1980’s there was an expansive survey that suggested around 25% of Latin America was at risk of contracting Chaga’s disease. This would be 100 million individuals. [6]
Approximately 12,000 people die from Chagas disease every year. [2] The United States is the country with the highest number of Chagas disease infected migrants. The country with the second highest number is Spain with an estimated 67,000 affected immigrants. [6] Due to the rising number of affected individuals in the United States, extra precautions have been implemented. For example, donated blood has been tested for Chagas disease since 2007. [4] The primary precautionary effort has been through screening blood donations. This practice has been legally implemented and enforced in almost all endemic countries such as Uruguay, Chile, and Brazil. Another preventative method is the use of insecticides. The incidence statistics indicate that the number of people newly infected every year has decreased drastically. This number went from around 700,000 in the year 1990 to just 41, 200 in 2006. The number of fatalities every year from Chagas disease has also decreased from 50,000 to 12,500 within the aforementioned years. [6]
Pathogenesis
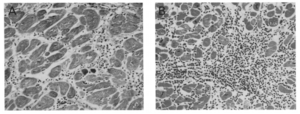
Chagas disease can also be passed on from mother to child, or to organ and blood recipients at any stage of the disease. [2] [6] Only 5% of pregnancies result in transmission of the parasite. [6] There are three stages of the disease: incubation, acute, and indeterminate. The incubation stage lasts for 7-15 days. Following this period, the acute stage lasts for several weeks. Finally, the indeterminate stage is believed to last indefinitely. Initial symptoms include the Romana sign which is a swelling of the eye. Infection can progress to megavisceras and heart disease following the indeterminate phase(Figure 3). Years following infection, symptoms can progress to Megavisceras in 8%, while heart disease is recorded in around 30%. [1] The remaining individual remain largely asymptomatic throughout their lives. [2] Immediately following of introduction of T. cruzi into the bloodstream, they infect cells, the location of reproduction. T. cruzi does not replicate in the bloodstream. These parasites reproduce by separating at the midgut and differentiate at the hindgut. [7] Cells infected by the parasite lyse, and the parasite spreads to other tissue and ganglion cells through the bloodstream and lymphatics. [6] Symptoms, if any, do not appear for one or two weeks following infection. [7]
Immediate symptoms to the bite are usually a small papule at the bite site. Other
symptoms include small inflammatory allergic reactions. This is uncommon an only occurs and
occurred in only 6.7% of a population studied in California. In some individuals, more drastic
symptoms, such as severe anaphylaxis, have been reported. In the United States, kissing bugs
are the most common biting bug to cause anaphylaxis [4] A compromised immune system, either due to immunosuppressant drugs or HIV/AIDS infection can result in the reactivation of Chagas disease [13] If the T-cells response is depressed, the infection becomes chronic. Damage and inflammation of the heart muscle are customary to the heart disease that results from Chagas disease. Ultimately, it can impair the dilation of all four chambers of the heart and the sinus node. It can also result in atrioventricular and intraventricular blocks. This can eventually lead to heart failure. Gastrointestinal disease may be caused by damage of the ganglia in the esophagus and colon. In the acute stage of infection, there are few symptoms. If a symptom occurs, it is typically a mild but prolonged fever. Other uncommon symptoms can include enlargement of the liver, spleen, and lymph nodes. [6]
Although it is known that organ tissue damage does occur, it is unknown whether this is due to direct interaction with the parasite or due to a factor of the immune response. [6] The immune response traditionally consists of B-cells, increased immunoglobulin in the blood, and anti-T.cruzi antibodies. Perhaps T. cruzi has been interacting with humans and other mammals for thousands of years, this immune response is generally ineffective. T. cruzi codes for surface proteins that lead to the coexpression of multiple antigens. These antigens are responsible for a delayed immune response. [7] As the immune response eventually grows stronger, the concentration of the parasite in the body is hindered to a degree. This marks the end of the acute phase of the infection. The remaining parasites, typically present in either muscle or ganglia, remain in the host indefinitely. [6]
Chronic Chagas’ disease Cardimyopathy causes heart lesions (CCC). It is thought that
CCC is caused by T-cells that recognize myosin proteins in the heart. An autoimmune response
was initially considered by researchers when the parasite could not be found near the heart
lesions. This autoimmune recognition occurs because T. cruzi antigen B13 and heart myosin
proteins are molecularly homologous. [14] There is a correlation between diabetes patients and those with T. cruzi infections. Hyperglycemia is thought to exacerbate T. cruzi infections. Studies have shown that patients suffering from Chagas disease tend to have insulin deficiencies. This is because adipose tissue serves as an idyllic environment for the parasite and there is an alteration of the level of adipokines that are released from the adipose tissue. This is concerning due to the increasing prevalence of Type II diabetes amongst Latin American countries. During the acute stage of the infection, patients are often hypoglycemic. This was found not be related to insulin levels but rather the increased glucose uptake in tissue cells by T. cruzi or decreased liver glucose production during acute T. cruzi infection. [13]
Treatment
Testing for Chagas disease can be complicated. Thus, the WHO recommends completing a minimum of two approved test before diagnosing an individual with the disease. This does not apply for blood screening where a singular test is sufficient. Approved diagnostic tests include the Chagatest recombinant v3.0 enzyme-linked immunoabsorbant assay (ELISA), an immunofluorescent antibody assay (IFA), and an antigen immunoblot (TESA IB). ELISA is FDA approved, while IFA and TESA IB are only used by the CDC and have yet to be approved by the FDA. Regarding blood screening, the T. cruzi EIA test system and PRISM Chagas chemiluminescent immunoassay are FDA approved options. [6] Treatment is typically only effective during the acute phase. It will both cure the infection and prevent any chronic expressions of Chagas’ disease. [6]
Treatment is “reasonable safe and effective” when implemented during the acute phase but significantly less so during the chronic phase. [2] [6] The reason why it is difficult to treat Chagas’ disease at the chronic stage is because it is difficult to find parasites at either peripheral blood or tissues. Even in the instance of a heart transplant, the disease persists. [24] Treatment options for T. cruzi include the use of benznidazole and nifurtimox. Neither drug has FDA approval but can be obtained through the CDC as per the protocol for disease investigation. [2] These drugs have regional differences in effectiveness. Nifurtimox was effective against the parasite during the acute stage for 80% of patients tested in regions across Chile, Argentian and Rio Grande do Sul. In other regions across Brazil, it was only effective amongst 40% of patients. This regional variation in treatments was not present for benznidazole. [24]
The side-effects of benznidazole are generally mild. Nausea, anorexia, headaches, stomach-aches, and joint pains were reactions among less than 5% of users. In a 60 day trial, benznidazole treatment of T. cruzi early in the chronic stage proved to be effective in 55.8% of patients [1]. There is currently no vaccine for Chagas disease; thus, most efforts remain preventative vector based.[6]
Additional drugs and compounds are currently being tested to find alternate treatments for Chagas disease. Research shows that aromatic diamidines, such as furamidine and its analogues can induce apoptosis in T. cruzi. These diamidines lead to DNA condensation and fragmentation in the nucleus, phosphatidylserine exposure, and diminished mitochondrial membrane potential in the parasite. The combination of these effects results in cell death. A drug has not yet been developed with diamidines and research continues on the mechanisms of action. It was also found that the venom of the snake Bothrops jararaca constrains the growth of the parasite in the epimastigotic stage by limiting mitochondrial processes. [18]
The in vitro infection of cells has been tested since the 1980’s. Macrophages make up the majority of the cells that are infected. For decades, the primary mechanism of infection was thought to be either phagocytosis or active penetration. It is now know to be endocytosis for bloodstream trypomastigotes and epimastigotes entering macrophages. Treatment of macrophages with cytochalasin B is known to decrease the internalization of epimastigotes by 98% and trypomastigotes by approximately 96%. In addition, cytochalasin B decreased the ability of the parasite to adhere to the surface of the macrophage. During the course of this particular experiment, no effect was noted on the parasites themselves. Macrophages were also unsuitable for parasite entrance if the phagocytes were kept at 4 degree Celsius before inoculation. Regarding mechanism, the parasites were still able to adhere to the surface of the macrophages, but not able to enter. [27]
One particular study found that macrophages increased their rate of trypomastigotes in mice when treated with trypsin. Other drugs that also increased macrophage ingestion were neuraminidase and periodate. [25] Studies also show that for the parasite to be prolific in host cells, Ca2+ mobilization is necessary. Increased expression of two surface glycoproteins ,82 kDa and 5/50 kDa, has been linked to suppressing Ca2+ signals. Thus, these glycoproteins have the ability to decrease the infectability of T. cruzi. [26]
During cell replication, T. cruzi is temporarily held at parasitophorous vacuoles. This is also where oxygen and nitrogen compounds are often synthesized. Trypanosomites utilize various antiooxidation systems to avoid cell damage during this time. The parasite employs cTXNPx and m-TXNPx, two enzymes that catalyze hydrogen peroxide reduction, to minimize interactions with hyperreactive molecules. Thus, peroxiredoxins, which are complete redox reactions, show potential for designing TXNPx inhibitors that may decrease the virulence of Chagas disease. [20] In an experiment conducted with rats, once a subject was cured of a T. cruzi infection, it was insusceptible to reinfection. In addition, injecting never infected rats with serum from the recovered rats conferred some protection. It only allowed for a partial development of the infection while serum injections continued. Once injections are halted, a relapse into the full potential of the infection occurs. When the parasites are embedded in somatic cells, the serum is no longer effective since only forms that live in the blood stream are vulnerable. [21]
In an experiment conducted with mice, male mice were more susceptible to T. cruzi infection than female mice. This applied only three strains studied during the experiment. [22] Thus, sex should be considered when controlling for human drug tests.
References
6. Rassi, Anis, and José Antonio Marin-Neto. "Chagas disease." The Lancet 375.9723 (2010): 1388-1402.
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2017, Kenyon College.
