User:S4371879
Jeniffer Denisse Loaiza Naranjo Bench D 31/08/16 [1]
Classification
Higher order taxa
Bacteria – Terrabcateria group – Actinobacteria – Actinobacteria – Micrococcales – Micrococcaceae – Rothia
Species
Rothia dentocariosa ATCC 17931
Description and significance
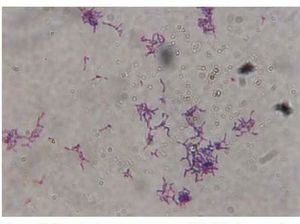
Rothia dentocariosa (R. dentocariosa) is a bacteria first described and isolated by George and Brown 1967 from human clinical samples of patients with caries [1]. It is a Gram-positive organisms, with a pleomorphic morphology. Its presentation can be as filamentous form with branches in solid environments or in cocci form in fluid environment (Figure 1) [2]. R. dentocariosa can be seen in clusters, individually or in chains. R. dentocariosa is commonly found in oral cavity of humans and correlated with periodontal diseases [2], [3]. Due to its prevalence and pathogenesis to humans, it is important to study this organism and its pathways for infection.
Genome structure
Select a strain for which genome information (e.g. size, plasmids, distinct genes, etc.) is available.
Rothia Dentocariosa ATCC 17931 (NC_014643) [4]. It has a circular DNA of size 2 506 025 bp. It has 2126 genes genes, 24 Pseudogenes and 2040 coding-sequences [4].
Cell structure and metabolism
R. dentocariosa is related to the Actinomyces species but it can be differentiated by its cell wall. Structurally, the cell wall does not possess DAP, instead the peptidoglycan is based in L-Lys–Ala3 bridges [2], [5]. The major sugars in the cell wall are galactose, fructose, glucose and ribose, while the fatty acid component consists of methyl-branched fatty acids, unsaturated menaquinones, Diphosphatidylglycerol (DPG) and Phoshatidylglycerol (PG) [2], [6]. Interestingly, dimannosyl diglyceride has higher prevalence in the filamentous from than the coccal form of R. dentocariosa [5]. The oral microbiome consists of many microbes that live in a community. These microbes tend to form biofilms to protect themselves from the immune system and have better chance of survival and virulence. R. dentocariosa coaggregates with Actinomyces species to form strong biofilms, Actinomyces spp. use Type 2-fimbrial adhesins [7].
.
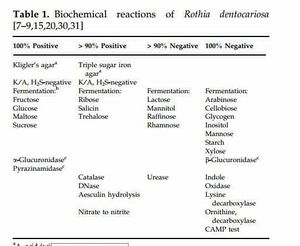
Some of the physiological characteristics of R. dentocariosa are the inability of producing spores, it does not have aerial micellium and has no motility. [1], [2]. Chemical tests in Table 1, help to identify this species more correctly. It reduces nitrate to ammonia, is catalase positive, oxidase and urease negative [2]. R dentocariosa can perform nitrate and nitrite reduction and decomposes casein [2]. It is a fermentative bacteria due to its usage of mannose, sucrose, fructose, maltose, glycerol and glucose to ferment lactic and acetic acid [2]. It cannot hydrolyse starch [2].
Ecology
R. dentocariosa is an aerobic bacteria but it can also grow in microaeophilic environments [2]. The best temperature to cultivate this bacteria is at 35° C [2]. The abundance of carbon, oxygen and nitrogen in the oral cavity makes it a comfortable environment for bacteria in general. Streptococci species are the most abundant followed by Haemophilus, Gemella, Rothia and Actinomyces species [8], [14]. Neisseria species and Rothia dentocariosa are one of the early colonizers of the oral cavity. This species appeared in the first 6 hour after colonization, commonly located in the sublingual sulcus [8].
One of the reasons that R. dentocariosa inhabits the oral cavity is a possible symbiotic interaction. Humans provide the bacteria with nutrients, and the bacteria produce nitrite. This can be further processed to nitric oxide, which acts as an antimicrobial agent [9]. Nitrite production is concentrated in the dorsal surface of the tongue [10]. Veilonella was the major group of nitrite production, followed by Actinomyces and in the third place were Rothia spp. [10]. Less caries were found in patients with high nitrate-reducing ability [10]. R. dentocariosa can reduce nitrate, which seems to be advantageous for the host. But still, R. dentocariosa has been reported to cause some pathologies to humans.
Pathology
R. dentocariosa causes inflammation in some patients, when the microbial environment is imbalanced. This inflammation is generally present in gingivitis and periodontitis [3], [11]. The immune response can be activated by various pathways. Expression of levan cause classical and alternate complement activation, which is related to cytotoxicity and may help the development of periodontal disease [12]. Additionally, R. dentocariosa has been reported to produce cytokines such as Tumor necrosis factor (TNF-α) [11]. This response is mediated by Toll-like receptors 2 (TLR2) generated by immune cells in the host, which is thought to generate the inflammation seen in gingivitis and periodontitis [3], [11].
R. dentocariosa predominated in patients with initial root caries lesions. Its prevalence in root surface was less than the initial root caries [13]. Interestingly, after the treatment this species decreased significantly and is was negatively related with bleeding in gingivitis [3], [13]. This seems to indicate that R. dentocariosa has a specific role in the intial steps of the colonization of the tooth [14]. Other study showed that R. dentocariosa and Scardovia inopinata are present in 50% of caries, while it is absent in caries-free subjects [3]. The pathogenesis of Rothia in oral diseases is not fully understood yet.
Additionally, R. dentocariosa rarely can cause systemic infection. The most common is endocarditis [15]. Most of the patients had a previous heart condition (85%) and some had a dental condition (60%) [15]. Some of the patients also had a related mycotic aneurysm (25%) [15]. If the antibiotic treatment did not work, the patients had to recur to surgery [15]. Rare infections can be spotted in other places. Lung cancer patients have been reported to have Pneumonia due to Rothia Dentocariosa [16]. Rarer cases of peritonitis [17], Bacteremia [18], Sepsis [19] tonsillitis [20] and prostheses infections [21], [22]. It generally affected immunosuppressed patients, but treatment with antibiotics was effective.
Application to biotechnology
Glutamine endopeptidase enzyme have been extracted from Rothia species, This enzyme have gliadin degrading activity and it is used to treat celiac disease. The major enzyme product is neprilysin (WP_005508036.1) extracted from Rothia mucilaginosa and Metalloendopeptidase PepO (WP_004006409.1) or Peptidase M13 (WP_048752476.1) from Rothia dentocariosa [4], [23]. Some embodiments used both species to generate the enzyme [23]. A BLAST search found 77% and 76% similarity between neprilysin and R. dentocariosa enzymes respectively.
Current research
The current research in this species is related to biofilm inhibition. R. dentocariosa found in biofilm surrounding silicone voice prostheses was treated with carboxymenethyl-chitosan and the reduction of the biofilm was of 23% less after 22 days. This antibiotic works in a long-time period [19]. Treatments with ZnO nanoparticles (35 nm) has been tested against biofilms and it resulted in inhibition of both R. dentocariosa and R. mucilaginosa with an IC50 of 53 μg/ml and 76 μg/ml respectively [10]. Lastly, another point of research is the research for specific antibodies and antigens, to generate a better immune response. As TLR2 was not the only receptor responsible by the host cells immune response, it was suggested that nucleotide binding oligomerization domain containing 2 (NOD2) protein could be the other receptor that participates in the immune response [11].
References
4. NCBI
6. Pandhi P, Hammond B. (1975) A glycolipid from Rothia dentocariosa. Arch Oral Biol 20: 399-401.
- ↑ MICR3004
This page is written by <Jeniffer Denisse Loaiza Naranjo> for the MICR3004 course, Semester 2, 2016
