Xylitol in Dental Decay Prevention
Dental decay is the demineralization of teeth caused by the fermentation of carbohydrates by bacteria such as Mutans streptococcus present in dental plaque. Xylitol is a sugar alcohol used as a sugar substitute which is not fermentable by plaque bacteria. It therefore plays a role in preventing dental decay.
Introduction
Dental infections of the teeth and gums are arguably the most common bacterial infections in humans. Dental plaque is the accumulation of bacterial communities in aggregates on the tooth surface. Of the 200-300 microbe species indigenous to dental plaque, only a select few are cariogenic, meaning they cause tooth decay. M. streptococcus is one of these cariogenic oral pathogens found in dental plaque. It produces extracellular glucans (polysaccharide chains of glucose monomers) allowing it to form aggregates on the tooth surface. The metabolism of fermentable carbohydrates by M. streptococcus produces lactic acid which causes a sharp drop in pH at the tooth surface (See figure 1). This increased acidity causes demineralization beneath the tooth surface which can then lead to cavity formation.[3]
Xylitol is a sugar alcohol used a non-sugar sweetener. Xylitol is non-cariogenic as well as cariostatic. Xylitol cannot be metabolized by M. streptococcus and therefore does not contribute to tooth demineralization. It has been found that chewing gum containing xylitol is effective in preventing dental caries compared with not chewing gum or chewing sugared gum.[3]
Dental Decay
Dental decay begins with subsurface demineralization causing the formation of a subsurface lesion known as a white spot followed by cavitation. Demineralization occurs whenever fermentable carbohydrate diffuses into plaque and is converted to acid end-products. Between meals pH levels return to normal and demineralization is repaired. Remineralization occurs when calcium and phosphate ions in the plaque diffuse into the lesion driven by the supersaturated concentration gradient. Demineralization leads to cavitation when the magnitude and frequency of acid production overwhelms the repair process. This can be caused by frequent eating or reduction of salivary flow.[3]
The tooth surface is both hard and non-shedding with an acquired enamel pellicle (AEP). The AEP is a membranous layer that protects the tooth from bacterial infection, formed when the tooth selectively absorbs acidic glycoproteins from the saliva. The AEP contains a high number of sulfate and carboxyl groups which gives it a net negative charge. Bacteria found in the saliva also have a net negative charge so there is a repulsion between the tooth surface and oral bacteria, which protects the tooth from infection. Dental plaque forms despite the fact that M. streptococcus is not a particularly effective colonizer. Patients exhibiting rampant caries have either frequent or excessive sucrose intake or low saliva flow, and are also found to have elevated levels of M. streptococcus in their saliva.[1]
Tooth Morphology
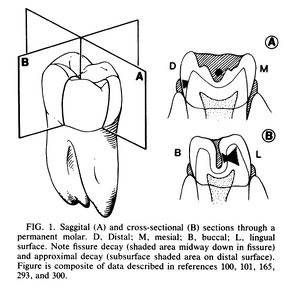
The morphology of the tooth dictates the bacterial composition of the plaque. The different ecosystems on the tooth favor some bacterial populations over others. Decay occurs primarily in the molars and premolars. The crown of the tooth, the visible portion of the tooth which is above the gumline, has five different surfaces upon which cariogenic plaque can form (See figure 1). The smooth surfaces on the cheek and tongue sides of the tooth are most prone to plaque formation but will only become decayed in special circumstances such as low saliva conditions or when substrate (carbohydrates) concentrations are high. The front and back of the tooth are also prone to plaque formation and are more prone to decay. In contrast, the chewing surface of the tooth is less prone to plaque formation but is the most prone to caries formation.[1]
Fissures and contact sites between teeth are the most caries prone parts of the tooth (see figure 1). Saliva is an important defense mechanism against dental decay for it’s buffering and remineralization abilities. Saliva stagnates at fissures and contact sites so it can not effectively buffer pH drops due to fermentation. Stagnation also prevents the replenishment of mineral lost from the tooth. Saliva can effectively inhibit M. streptococcus growth by buffering the environment because M. streptococcus is an aciduric bacteria, meaning it thrives in acidic conditions.[1]
Virulence Factors
Fermentable Carbohydrates
M. streptococcus levels in plaque increase in the presence of fermentable carbohydrates such as sucrose. When sucrose is restricted, the rate of dental decay and the plaque levels of M. streptococcus decrease. When sucrose is in excess there is an increase in lactic acid, a product of the fermentation of sucrose. Of the sucrose that enters the cell, a small portion is transformed into glucans. These glucans either remain in the cell or diffuse into the environment. Glucans act as adhesive material which allow M. streptococcus to form adhesive colonies on the tooth surface. In smooth surface decay, glucan formation is a key virulence factor.[1]
pH
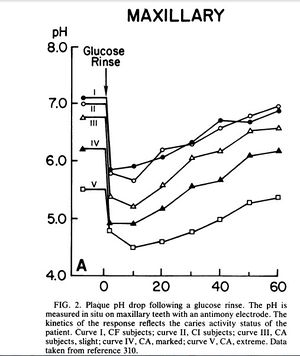
Following exposure to sucrose or other fermentable carbohydrates, the pH in plaque can drop below 5.0 for anywhere from 30 - 120 minutes (See figure 2). Most plaque bacteria are not metabolically active at such a low pH, therefore M. streptococcus is selected for in these niches because it is aciduric with a pH optima of 5.0 - 5.5. In times of nutrient excess an intracellular polysaccharide is formed by plaque bacteria which keeps the plaque ecosystem below pH 5.5 for extended lengths of time after carbohydrate exposure. This is another virulence factor in dental decay correlated with caries activity and carbohydrate consumption.[1]
A study examined the effect of a glucose rinse on the pH levels of subjects with varying degrees of cavitation (see figure 2). The pH was read at periodic intervals until it returned to its original level. The rapid drop in pH shows that glucose was instantaneously converted to acid products, mainly lactic acid, which overwhelmed the buffering capabilities of saliva. In subjects with active dental caries the pH stayed below 5.0 for 20 - 50 minutes while in caries-free or caries-inactive subjects the pH dropped to around 6.0 and returned to normal after 40 minutes. Further analysis showed that the plaque present at caries-active sites produced twice the acid per milligram than the plaque at caries free sites.[1]
Decay Prevention
Remineralization can be encouraged if plaque acid production is restricted through a low sucrose diet, fluoride treatment, or use of sugar substitutes such as xylitol.[1] Chewing sugar-free or xylitol-sweetened gum can also encourage remineralization as it increases salivary flow whose buffering capacity and high concentration of calcium and phosphate ions remineralize dental enamel preventing caries development.
[5]
Saliva
Saliva provides critical defense against dental decay. When food is chewed salivary flow increases providing a large liquid volume for plaque acids to diffuse into. These acids are neutralized by the bicarbonate buffer present in high concentrations in saliva. Saliva is also supersaturated with calcium and phosphate ions providing a constant remineralization mechanism. Additionally, salivary proteins and glycoproteins such as lysozyme, lactoperoxidase, lactoferrin, and high-molecular weight agglutinins, exhibit antibacterial activity.[1]
Fluoride
Fluoride is a common preventative treatment of dental decay by inhibiting the metabolic activity of bacteria. The low pH characteristic of carbohydrate fermentation magnifies it’s antimicrobial effect by facilitating the uptake of fluoride by the cell. Sugar is transported into the cell by a proton motive force generated by the pH gradient across the membrane. Fluoride is in the form of hydrogen fluoride (HF), which when diffused into the cell at a low pH releases enough hydrogen ions to lower the cytoplasmic pH below the optima of most cytoplasmic enzymes in plaque bacteria. This release of hydrogen ions also dissipates the pH gradient fueling the proton motive force thereby starving the bacterial cell by inhibiting substrate uptake.[1]
Xylitol and Dental Decay Prevention
Xylitol is a non-cariogenic non-sugar sweetener. Regular consumption of xylitol causes a shift in oral bacteria populations favoring less cariogenic bacteria.[5] The main side effect of polyols is osmotic diarrhea although 4-5 times the amount needed to prevent dental caries would need to be ingested for this to occur. It has been concluded that polyols including xylitol are non-cariogenic. Unlike other sugar alcohols, such as sorbitol, xylitol has been shown to protect and reduce tooth decay by reducing M. streptococcus levels in plaque and saliva thereby reducing lactic acid production. One study showed that groups consuming 100% xylitol had greater reduction in caries and M. streptococcus levels than groups which consumed a combination of xylitol and sorbitol.[4]
Mechanism of Action
Unlike sucrose, xylitol has a minimal effect on plaque pH.[4] When plaque is exposed to xylitol, concentrations of ammonia and basic amino acids increase, neutralizing plaque acids. [5] Xylitol is absorbed and accumulated in M. streptococcus. It competes with sucrose for cell wall transporters and metabolic processes. The cell expends energy breaking down xylitol with no energy yield and energy-producing intermediates are consumed but not reproduced. This inhibits the growth of M. streptococcus. When xylitol is consumed habitually long-term less virulent strains of M. streptococcus are selected for.[4] Xylitol also may affect the adhesive and cohesive properties of plaque which decreases the overall quantity of M. streptococcus in dental plaque. Some strains of M. streptococcus take up xylitol and convert it to xylitol-5-phosphate which results in the development of intra-cellular vacuoles and degraded cell members. This has a bacteriostatic effect on plaque bacteria.[5]
A study compared the growth of M. streptococcus strain OMZ 176 in the presence of xylitol, sorbitol, or glucose in culture media. It was found that 5% of xylitol in the growth medium inhibited growth of M. streptococcus compared to media with no extra carbon source added. When xylitol and glucose was added growth was reduced compared with glucose alone. Growth inhibition was dose related. Conversely, sorbitol did not show similar effects. In the presence of xylitol, the dental plaque formed was less adhesive and hydrated, and also contained less glucan and lipoteichoic acid than plaque grown in the presence of sucrose.[6]
Dosing
The recommended dosage and usage frequency of xylitol has yet to be concluded. Based on the current research 6 - 10 g divided into at least 3 consumptions per day is necessary to have an effect. One study found that the amount of xylitol consumed determined the degree of reduction in dental decay although the benefits of xylitol top out after 10 g per day. Any less than 5 g of xylitol per day is no more effective than sorbitol. It was concluded that other sugar alcohols may enhance the efficacy of xylitol but will not diminish it. Frequency of ingestion was also found to be important. Any less than 3 doses per day may not be effective.[4] Many studies have shown that xylitol-containing gum reduces the extent of dental caries. Candy, toothpaste, tablets, and snack foods are being developed and tested.[4]
Xylitol in Reducing Caries Risk in Children
Bacteriological studies have shown that the salivary levels of M. streptococcus in mothers is related to the initial acquisition in their infants. The earlier the colonization of Mutans streptococcus in children, the higher the caries rates later in life. This study looked at whether mothers chewing xylitol-sweetened gums could help to prevent caries in their children during tooth eruption.
After three months, the number of mothers with high M. streptococcus levels decreased. These mothers also showed an overall decrease in dental plaque. The frequency of xylitol consumption was correlated with the level of decrease in salivary M. streptococcus levels. A reduction in the mother’s salivary levels significantly affected the levels in her child. The study concluded that xylitol consumption significantly reduced salivary levels in both mothers and children and reduced plaque accumulation on the tooth surface in children. Therefore, children who adopt the habit of regularly chewing xylitol-sweetened gum can reduce caries risk.
[2]
Conclusion
Xylitol has been shown to be the most effective in preventing dental decay and improving oral health due to it's noncariogenicity. Regular exposure to xylitol (at least three times per day) can alter the bacterial population of the oral cavity, reducing the amount of M. streptococcus levels in both dental plaque and saliva. Candies, chewing gums, and supplements sweetened with xylitol are currently being developed as an accessible and affordable means to prevent dental decay. Like fluoride treatments, xylitol could be a widely accessible and commonly used means to improve dental health.
References
[6] Vegarud, Azzev. "Growth Inhibition of Streptococcus Mutans Strain OMZ176 by Xylitol." Acta Pathologica Et Micriobioligica Scandinavica I88.88 (1980): 61-63. Print.
[7]Wikipedia. Wikimedia Foundation, n.d. Web. 26 Apr. 2013.
Edited by (Zoe Kiklis), a student of Nora Sullivan in BIOL187S (Microbial Life) in The Keck Science Department of the Claremont Colleges Spring 2013.