CTXφ Bacteriophage: Difference between revisions
| Line 11: | Line 11: | ||
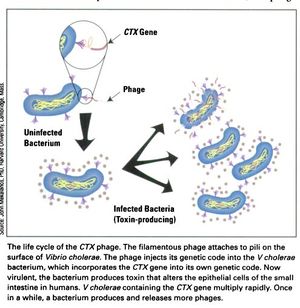
[[Image:CTX_Phage_life_cycle.jpg|thumb|right|<b>Figure 3:</b> The life cycle of the CTXφ Bacteriophage with <i>Vibrio cholerae</i> as its host.]] | [[Image:CTX_Phage_life_cycle.jpg|thumb|right|<b>Figure 3:</b> The life cycle of the CTXφ Bacteriophage with <i>Vibrio cholerae</i> as its host.]] | ||
In a process known as lysogenic phage conversion, the CTXφ bacteriophage integrates, among others, its <i>ctxAB</i> genes into its host, <i>Vibrio cholerae</i>. The <i>ctxAB</i> genes code for a type of exotoxin, called Cholera toxin or just "CT," that causes <i> V. cholerae</i> to switch from being nonpathogenic to highly virulent. For those infected, it is also the primary cause of the large amounts of watered-down diarrhea—cholera's main symptom.<ref name = Boyd>Boyd, E. F. (2010). "Efficiency and specificity of CTXphi chromosomal integration: dif makes all the difference." Proceedings of the National Academy of Sciences of the United States of America, 107(9), 3951–3952. https://doi.org/10.1073/pnas.1000310107</ref> After finding a live host and undergoing CTXφ particle adsorption (see Figure 3 at right) to the <i>V. cholerae</i> cell wall, viral single-stranded DNA is then injected into the cell cytoplasm and a complementary strand of DNA is synthesized in order to form a circular plasmid, pCTX,<ref name = Waldor>Waldor, M., Davis, B. M. (2003). "Filamentous phages linked to virulence of Vibrio cholerae," <i>Current Opinion in Microbiology,Volume 6, Issue 1</i>, Pages 35-42, ISSN 1369-5274. https://doi.org/10.1016/S1369-5274(02)00005-X. (http://www.sciencedirect.com/science/article/pii/S136952740200005X)</ref>which then integrates into the <i>V. cholerae</i> genome at a site-specific attachment location on one or both chromosomes of <i> V. cholerae</i>.<ref name = Boyd>Boyd, E. F. (2010). "Efficiency and specificity of CTXphi chromosomal integration: dif makes all the difference." Proceedings of the National Academy of Sciences of the United States of America, 107(9), 3951–3952. https://doi.org/10.1073/pnas.1000310107</ref> Because the CTXφ phage, like all bacteriophages, only injects its genome, or, more commonly, its <i>prophage</i>, the CTXφ integration makes use of many of the host cell's enzymes. An integrase enzyme is not present in the CTXφ genome, so it recruits XerC and XerD, two tyrosine recombinases present in <i>V. cholerae</i>, to catalyze this merge.<ref name = Boyd>Boyd, E. F. (2010). "Efficiency and specificity of CTXphi chromosomal integration: dif makes all the difference." Proceedings of the National Academy of Sciences of the United States of America, 107(9), 3951–3952. https://doi.org/10.1073/pnas.1000310107</ref> Normally, XerC and XerD fix chromosome dimers in eubacteria.<ref name = Sherratt>Sherratt, D. J., Søballe, B., Barre, F. X., Filipe, S., Lau, I., Massey, T., & Yates, J. (2004). "Recombination and chromosome segregation." Philosophical transactions of the Royal Society of London. Series B, <i>Biological sciences</i>, 359(1441), 61–69. https://doi.org/10.1098/rstb.2003.1365</ref> CTXφ prophages usually integrate as tandem prophages,<ref name = McLeod>McLeod, S. M., Kimsey, H. H., Davis, B. M., Waldor, M. K. (2005). "CTXφ and Vibrio cholerae: exploring a newly recognized type of phage–host cell relationship." Microreview, 57 (2), 347–356. https://onlinelibrary.wiley.com/doi/full/10.1111/j.1365-2958.2005.04676.x.</ref> getting replicated whenever the host cell's genome does. After sufficient time has passed and enough proteins and genetic material has been packaged, he phage proteins are localized to the inner membrane prior to forming virions.<ref name = McLeod>McLeod, S. M., Kimsey, H. H., Davis, B. M., Waldor, M. K. (2005). "CTXφ and Vibrio cholerae: exploring a newly recognized type of phage–host cell relationship." Microreview, 57 (2), 347–356. https://onlinelibrary.wiley.com/doi/full/10.1111/j.1365-2958.2005.04676.x.</ref> Like many other filamentous phages, once emergence from the host cell occurs, it does not require lysis of the host's membrane. It is more of a secretion.<ref name = Faruque>Faruque, S. M., & Mekalanos, J. J. (2012). "Phage-bacterial interactions in the evolution of toxigenic <i>Vibrio cholerae</i>." <i>Virulence</i>, 3(7), 556–565. https://doi.org/10.4161/viru.22351</ref> Secretion of CTXφ phages from the bacterial cell occurs through a host‐encoded channel, EpsD.<ref name = McLeod>McLeod, S. M., Kimsey, H. H., Davis, B. M., Waldor, M. K. (2005). "CTXφ and Vibrio cholerae: exploring a newly recognized type of phage–host cell relationship." Microreview, 57 (2), 347–356. https://onlinelibrary.wiley.com/doi/full/10.1111/j.1365-2958.2005.04676.x.</ref> | In a process known as lysogenic phage conversion, the CTXφ bacteriophage integrates, among others, its <i>ctxAB</i> genes into its host, <i>Vibrio cholerae</i>. The <i>ctxAB</i> genes code for a type of exotoxin, called Cholera toxin or just "CT," that causes <i> V. cholerae</i> to switch from being nonpathogenic to highly virulent. For those infected, it is also the primary cause of the large amounts of watered-down diarrhea—cholera's main symptom.<ref name = Boyd>Boyd, E. F. (2010). "Efficiency and specificity of CTXphi chromosomal integration: dif makes all the difference." Proceedings of the National Academy of Sciences of the United States of America, 107(9), 3951–3952. https://doi.org/10.1073/pnas.1000310107</ref> After finding a live host and undergoing CTXφ particle adsorption (see Figure 3 at right) to the <i>V. cholerae</i> cell wall, viral single-stranded DNA is then injected into the cell cytoplasm and a complementary strand of DNA is synthesized in order to form a circular plasmid, pCTX,<ref name = Waldor>Waldor, M., Davis, B. M. (2003). "Filamentous phages linked to virulence of Vibrio cholerae," <i>Current Opinion in Microbiology,Volume 6, Issue 1</i>, Pages 35-42, ISSN 1369-5274. https://doi.org/10.1016/S1369-5274(02)00005-X. (http://www.sciencedirect.com/science/article/pii/S136952740200005X)</ref>which then integrates into the <i>V. cholerae</i> genome at a site-specific attachment location on one or both chromosomes of <i> V. cholerae</i>.<ref name = Boyd>Boyd, E. F. (2010). "Efficiency and specificity of CTXphi chromosomal integration: dif makes all the difference." Proceedings of the National Academy of Sciences of the United States of America, 107(9), 3951–3952. https://doi.org/10.1073/pnas.1000310107</ref> Because the CTXφ phage, like all bacteriophages, only injects its genome, or, more commonly, its <i>prophage</i>, the CTXφ integration makes use of many of the host cell's enzymes. An integrase enzyme is not present in the CTXφ genome, so it recruits XerC and XerD, two tyrosine recombinases present in <i>V. cholerae</i>, to catalyze this merge.<ref name = Boyd>Boyd, E. F. (2010). "Efficiency and specificity of CTXphi chromosomal integration: dif makes all the difference." Proceedings of the National Academy of Sciences of the United States of America, 107(9), 3951–3952. https://doi.org/10.1073/pnas.1000310107</ref> Normally, XerC and XerD fix chromosome dimers in eubacteria.<ref name = Sherratt>Sherratt, D. J., Søballe, B., Barre, F. X., Filipe, S., Lau, I., Massey, T., & Yates, J. (2004). "Recombination and chromosome segregation." Philosophical transactions of the Royal Society of London. Series B, <i>Biological sciences</i>, 359(1441), 61–69. https://doi.org/10.1098/rstb.2003.1365</ref> CTXφ prophages usually integrate as tandem prophages,<ref name = McLeod>McLeod, S. M., Kimsey, H. H., Davis, B. M., Waldor, M. K. (2005). "CTXφ and Vibrio cholerae: exploring a newly recognized type of phage–host cell relationship." Microreview, 57 (2), 347–356. https://onlinelibrary.wiley.com/doi/full/10.1111/j.1365-2958.2005.04676.x.</ref> getting replicated whenever the host cell's genome does. After sufficient time has passed and enough proteins and genetic material has been packaged, he phage proteins are localized to the inner membrane prior to forming virions.<ref name = McLeod>McLeod, S. M., Kimsey, H. H., Davis, B. M., Waldor, M. K. (2005). "CTXφ and Vibrio cholerae: exploring a newly recognized type of phage–host cell relationship." Microreview, 57 (2), 347–356. https://onlinelibrary.wiley.com/doi/full/10.1111/j.1365-2958.2005.04676.x.</ref> Like many other filamentous phages, once emergence from the host cell occurs, it does not require lysis of the host's membrane. It is more of a secretion.<ref name = Faruque>Faruque, S. M., & Mekalanos, J. J. (2012). "Phage-bacterial interactions in the evolution of toxigenic <i>Vibrio cholerae</i>." <i>Virulence</i>, 3(7), 556–565. https://doi.org/10.4161/viru.22351</ref> Secretion of CTXφ phages from the bacterial cell occurs through a host‐encoded channel, EpsD.<ref name = McLeod>McLeod, S. M., Kimsey, H. H., Davis, B. M., Waldor, M. K. (2005). "CTXφ and Vibrio cholerae: exploring a newly recognized type of phage–host cell relationship." Microreview, 57 (2), 347–356. https://onlinelibrary.wiley.com/doi/full/10.1111/j.1365-2958.2005.04676.x.</ref> | ||
<br> | <br> | ||
Revision as of 04:04, 12 December 2020
Overview
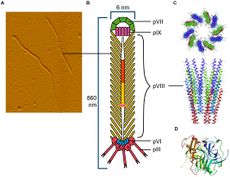
The CTXφ bacteriophage (or sometimes written as CTXphi bacteriophage) is a lysogenic, filamentous, single-stranded DNA (ssDNA) phage that is responsible for turning the previously non-infectious Vibrio cholerae into a highly pathogenic microbe that causes cholera in humans.[2][3][4] This particular bacteriophage,Vibrio virus CTXphi belongs to the family Inoviridae and the genus Affertcholeramvirus. These phages are composed of only 5 viral proteins and are between 1 and 2 microns in length.[5] They resemble something like a cooked spaghetti noodle. (See Figure 1 at right).
Genetic Material
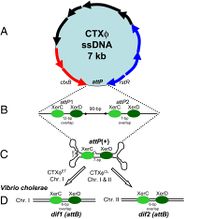
The CTXφ phage can be identified in its host in both its replicative form (when the bacteriophage is in lysogeny, or the lytic cycle), and in its non-replicative form, which is most common. It is in fact extremely rare to find or identify a CTXφ bacteriophage outside a host. This ssDNA phage, when in its lysogenic cycle, integrates its own genetic material in such a way as to form the most stable lysogens.[4] The average size of a CTXφ genome is about 6.9 to 7 kb long and consists of two sections; the RS2 region and the core region.[6] The core contains the genes that code for the Cholera toxin.[6]
Infection, Replication & Release from Host Cell
In a process known as lysogenic phage conversion, the CTXφ bacteriophage integrates, among others, its ctxAB genes into its host, Vibrio cholerae. The ctxAB genes code for a type of exotoxin, called Cholera toxin or just "CT," that causes V. cholerae to switch from being nonpathogenic to highly virulent. For those infected, it is also the primary cause of the large amounts of watered-down diarrhea—cholera's main symptom.[4] After finding a live host and undergoing CTXφ particle adsorption (see Figure 3 at right) to the V. cholerae cell wall, viral single-stranded DNA is then injected into the cell cytoplasm and a complementary strand of DNA is synthesized in order to form a circular plasmid, pCTX,[7]which then integrates into the V. cholerae genome at a site-specific attachment location on one or both chromosomes of V. cholerae.[4] Because the CTXφ phage, like all bacteriophages, only injects its genome, or, more commonly, its prophage, the CTXφ integration makes use of many of the host cell's enzymes. An integrase enzyme is not present in the CTXφ genome, so it recruits XerC and XerD, two tyrosine recombinases present in V. cholerae, to catalyze this merge.[4] Normally, XerC and XerD fix chromosome dimers in eubacteria.[8] CTXφ prophages usually integrate as tandem prophages,[9] getting replicated whenever the host cell's genome does. After sufficient time has passed and enough proteins and genetic material has been packaged, he phage proteins are localized to the inner membrane prior to forming virions.[9] Like many other filamentous phages, once emergence from the host cell occurs, it does not require lysis of the host's membrane. It is more of a secretion.[10] Secretion of CTXφ phages from the bacterial cell occurs through a host‐encoded channel, EpsD.[9]
CT & non-CT Toxins
The CTXφ phage contains both types of exotoxins.
CT Toxins
Non-CT Toxins
Recent research suggests that there are perhaps two toxins (other than CT) are produced from genes of the CTXφ genome. The first of these is accessory cholera enterotoxin, or Ace. Ace is currently thought to be a minor coat protein of CTXφ, though the process by which the toxin could be released from the protein coat is not yet known to scientists. The second non-CT toxin encoded within the CTXφ bacteriophage genome is zonula occludens toxin, or Zot. Zot, though absolutely essential for the production of the CTXφ virion, is not actually present in the phage particle. The part that Ace and Zot play in the virulence of cholera is still unknown.[7]
Conclusion
Overall text length should be at least 1,000 words (before counting references), with at least 2 images. Include at least 5 references under Reference section.
References
- ↑ Gagic, D., Ciric M., Wen W., Ng F., Rakonjac J. (2016). "Exploring the Secretomes of Microbes and Microbial Communities Using Filamentous Phage Display." Frontiers in Microbiology, 7:429. https://doi.org/10.3389/fmicb.2016.00429.
- ↑ Davis, B. M., Kimsey, H. H., Chang, W., & Waldor, M. K. (1999). "The Vibrio cholerae O139 Calcutta bacteriophage CTXφ is infectious and encodes a novel repressor." Journal of Bacteriology, 181(21), 6779-6787. https://www.frontiersin.org/articles/10.3389/fmicb.2016.00429/full
- ↑ Ochman, H., Lawrence, J. & Groisman, E. (2000). "Lateral gene transfer and the nature of bacterial innovation." Nature, 405, 299–304. https://doi.org/10.1038/35012500.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 Boyd, E. F. (2010). "Efficiency and specificity of CTXphi chromosomal integration: dif makes all the difference." Proceedings of the National Academy of Sciences of the United States of America, 107(9), 3951–3952. https://doi.org/10.1073/pnas.1000310107
- ↑ Mai-Prochnow, A., Hui, J., Kjelleberg, S., Rakonjac, J., McDougald, D., Rice, S. (2015). "Big things in small packages: the genetics of filamentous phage and effects on fitness of their host." FEMS Microbiology Reviews, Volume 39, Issue 4, Pages 465–487, https://doi.org/10.1093/femsre/fuu007
- ↑ 6.0 6.1 Fan, F., Kan, B.(2015). "Survival and proliferation of the lysogenic bacteriophage CTXφ in Vibrio cholerae." Virologica Sinica. 30, 19–25. https://doi.org/10.1007/s12250-014-3550-7
- ↑ 7.0 7.1 Waldor, M., Davis, B. M. (2003). "Filamentous phages linked to virulence of Vibrio cholerae," Current Opinion in Microbiology,Volume 6, Issue 1, Pages 35-42, ISSN 1369-5274. https://doi.org/10.1016/S1369-5274(02)00005-X. (http://www.sciencedirect.com/science/article/pii/S136952740200005X)
- ↑ Sherratt, D. J., Søballe, B., Barre, F. X., Filipe, S., Lau, I., Massey, T., & Yates, J. (2004). "Recombination and chromosome segregation." Philosophical transactions of the Royal Society of London. Series B, Biological sciences, 359(1441), 61–69. https://doi.org/10.1098/rstb.2003.1365
- ↑ 9.0 9.1 9.2 McLeod, S. M., Kimsey, H. H., Davis, B. M., Waldor, M. K. (2005). "CTXφ and Vibrio cholerae: exploring a newly recognized type of phage–host cell relationship." Microreview, 57 (2), 347–356. https://onlinelibrary.wiley.com/doi/full/10.1111/j.1365-2958.2005.04676.x.
- ↑ Faruque, S. M., & Mekalanos, J. J. (2012). "Phage-bacterial interactions in the evolution of toxigenic Vibrio cholerae." Virulence, 3(7), 556–565. https://doi.org/10.4161/viru.22351
Edited by Tara Cerny, student of Joan Slonczewski for BIOL 116 Information in Living Systems, 2019, Kenyon College.