Flooded Soils: Difference between revisions
Amsutherland (talk | contribs) |
Jlchiartas (talk | contribs) m (Jlchiartas moved page Central Metabolism (Flooded soils) to Flooded Soils) |
||
| (133 intermediate revisions by 7 users not shown) | |||
| Line 1: | Line 1: | ||
{{Curated}} | |||
[[Image:Floridacrocodile1.jpg|thumb|500px|right|Ding Darling reserve, Sanibel Island, Florida, with an American Crocodile. Wikipedia jimfbleak 13:37, 2 April 2006 (UTC)]] | |||
==Introduction== | ==Introduction== | ||
[[Image:flooded soil.png|thumb|400px|schematic presentation of flooded soil from[[Y.Chen and Y Avnimelech]]]] | |||
[[Image:flooded soil.png|thumb|schematic presentation of flooded soil from[[Y.Chen and Y Avnimelech]]]] | |||
'''Flooded soils''' occur with complete water saturation of soil pores, and generally result in anoxic conditions of the soil environment. Flooded soil environments may include such [[Wikipedia:ecosystem|ecosystem]] | '''Flooded soils''' occur with complete water saturation of soil pores, and generally result in anoxic conditions of the soil environment. Flooded soil environments may include such [[Wikipedia:ecosystem|ecosystem]] | ||
as: rice paddies; wetlands (swamps, marshes, and bogs); compacted soils; and post-rain soils (Scow, 2008). Additionally, similar redox conditions (where oxygen is lacking) can also be found within soil aggregates and along pollutant plumes, and thus many of the concepts discussed in this section may be applied to those environments. | as: rice paddies; wetlands (swamps, marshes, and bogs); compacted soils; and post-rain soils (Scow, 2008). Additionally, similar redox conditions (where oxygen is lacking) can also be found within soil aggregates and along pollutant plumes, and thus many of the concepts discussed in this section may be applied to those environments. | ||
| Line 14: | Line 15: | ||
[[Image:phenomenon in aggregates.png|thumb|left|200px|Electron acceptor used in aggregates. adjusted from [[Prof. Kate lec #5]]]] | [[Image:phenomenon in aggregates.png|thumb|left|200px|Electron acceptor used in aggregates. adjusted from [[Prof. Kate lec #5]]]] | ||
[[Image:phenomena in pollutant plume.png|thumb|200px|Order of electron acceptor in pollutant plume from [[USGS]]]] | [[Image:phenomena in pollutant plume.png|thumb|200px|Order of electron acceptor in pollutant plume from [[USGS]]]] | ||
In general, flooded | In general, flooded soils occur due to seasonal flooding or agricultural activity. | ||
Flooded soils can be often converted into non-flooded soils by the water level fluctuation and drainage. Through this variation of soil conditions, various gases are emitted into the atmosphere and environmental factors, such as redox potential (Eh), pH, acidity, alkalinity, and salinity, are continuously changing. | |||
As explained in the | As explained in the [[#Introduction]], microorganisms can use alternative terminal electron acceptors (such as nitrate, perchlorate, sulfate, and carbon dioxide) when dissolved oxygen is absent. Microbes will successively use electron acceptors according to the order of energy yields resulting from electron acceptor utilization indicted on the electron tower (see [[#Electron Tower]] theory). The progression of electron acceptor utilization may also be observed in soil aggregates and pollutant plumes. | ||
===[http://en.wikipedia.org/wiki/Redox Oxidation/Reduction (Redox) Reaction]=== | |||
In redox reactions, one molecule (the reducing agent) loses electrons and another molecule (the oxidizing agent) accepts electrons. A classic example well known in the process of cellular respiration is when glucose (the reducing agent) reacts with oxygen (the oxidizing agent)and is oxidized to carbon dioxide. In this reaction, oxygen is reduced to water. Oxygen is the most common and highest energy yielding electron acceptor, and some organisms (strict aerobes) can not live long without it.(6) In flooded soils oxygen is typically not available. Facultative and strict anaerobic bacteria have the ability to use other oxidizing agents/electron acceptors to carry out respiration. Anaerobic and facultative bacteria will use the electron acceptor which yields the highest energy, or the acceptor which is most readily available. The availability and concentration of electron acceptors changes as the soil profile increases in depth. | |||
====Electron Tower==== | |||
[[Image:Environmental1.gif|thumb|350px|Electron tower [[http://www.microbiologybytes.com/introduction/Environmental.html]]]] | |||
Electron tower theory explains the utilization order of electron acceptor for respiration. | |||
Depending on the type of electron acceptors used by microorganisms, microbes can be classified as strict aerobes, obligate anaerobes, and facultative anaerobes. Strict aerobes can not live under anoxic condition; on the contrary, obligate anaerobes can never use oxygen as electron acceptor. However, facultative anaerobes can live in both aerobic and anaerobic condition. If oxygen is plentiful, they tend to use oxygen because microorganisms gain much energy from reducing oxygen rather than other electron acceptors. When there is no more available oxygen in solution, microbes will choose to use nitrate as an electron acceptor (if available). Thus, obligate anaerobes and facultative anaerobes use alternative electron acceptors in the order of electron acceptor having the most reducing energy. Oxygen is the most efficient electron acceptor, while carbon dioxide has the least amount of reduction potential. | |||
====Gleyed Soils and Recovery to Aerobic Conditions==== | |||
[[Image:Gleyed soil.png|thumb|200px|left|Gleyed soil from Prof. Scow's lecture note 2008]] | |||
[[Image:Oxidized soil.png|thumb|300px|Oxidized soil from Prof. Scow's lecture note 2008]] | |||
[[Image:Winogradsky column.jpeg|thumb|300px|right|[http://dwb.unl.edu/Teacher/NSF/C11/C11Links/helios.bto.ed.ac.uk/bto/microbes/winograd.htm Winogradsky column ]]] | |||
'''Soil Gleying''': | |||
Gleying is a phenomenon in which waterlogged soils are discolored by accumulation of Fe(II) due to reduction of ferric iron into ferrous iron (Lovely 1991). | |||
Although ferric iron exists as an insoluble form in flooded soils, more ferrous iron can accumulate by the reduction of ferric iron over time. This results in a greenish, blue, grey soil color. In general Fe(III)-reducing fermentative bacteria can be readily isolated from gleyed soils. | |||
The black color of soils/solution is frequently observed in flooded soil. This may result from the formation of iron sulfides (FeS) and pyrite (FeS<sub>2</sub>) (Wenk and Bulakh 2004). | |||
'''Recovery to Aerobic Conditions''' | |||
When waterlogged soils drain, the Eh starts to increase as oxygen diffuses into soil pores. Plentiful oxygen represses the activity of anaerobes, which results in an increase of aerobic microbes. If oxygen diffuses deep into the soil profile, the production of H<sub>2</sub>S ceases. Under aerobic conditions, ferrous iron is oxidized by iron-oxidizing bacteria, resulting in the formation of ferric oxides or ferric hydroxide minerals. The gray color in soil changes to a red, yellow, or brown color as these minerals are oxidized. At higher Eh zones ( > 500 mV), undecomposed soil organic matter is used as an electron donor by aerobes and converted to water and CO<sub>2</sub> (Richardson and Vepraskas 2000). | |||
In aerobic condition, manganese can be readily oxidized to MnO<sub>2</sub>. Black soil is often shown in aerated soil because manganese oxide is a mineral with a black color. | |||
When | |||
===Variation of pH and Eh=== | ===Variation of pH and Eh=== | ||
| Line 34: | Line 44: | ||
Neutral pH soil | Neutral pH soil | ||
When soil is saturated with water, pH drops at first due to organic acid produced from fermentation. Then, pH gradually starts to rise | When soil is saturated with water, pH drops at first due to organic acid produced from fermentation. Then, pH gradually starts to rise as a result of the buffering capacity of the soil. The half reactions of hydrogen consumption are as follow; | ||
Aerobic respiration: ½ O<sub>2</sub> + 2e<sup>-</sup> + 2H<sup>+</sup> -> H<sub>2</sub>O (by facultative | Aerobic respiration: ½ O<sub>2</sub> + 2e<sup>-</sup> + 2H<sup>+</sup> -> H<sub>2</sub>O (by facultative anaerobes and aerobes) | ||
Denitrification: 2NO<sub>3</sub><sup>-</sup> + 12 H<sup>+</sup> +10e<sup>-</sup> -> N<sub>2</sub>+6H<sub>2</sub>O (by denitrifiers) | |||
Manganese reduction: MnO<sub>2</sub> + 4H<sup>+</sup> + 2e<sup>-</sup> ->Mn<sup>2+</sup> + 2H<sub>2</sub>O (by manganese reducing bacteria) | |||
Iron reduction: Fe(OH)<sub>3</sub> + 3 H<sup>+</sup> + 2e<sup>-</sup> -> Fe<sup>2+</sup> + 2H<sub>2</sub>O (by iron reducing bacteria) | |||
Sulfate reduction: SO<sub>4</sub><sup>2-</sup> + 10H<sup>+</sup> +8e<sup>-</sup> -> H<sub>2</sub>S + 4H<sub>2</sub>O (by sulfate reducing bacteria) | Sulfate reduction: SO<sub>4</sub><sup>2-</sup> + 10H<sup>+</sup> +8e<sup>-</sup> -> H<sub>2</sub>S + 4H<sub>2</sub>O (by sulfate reducing bacteria) | ||
Methane production: CO<sub>2</sub> + 8 H<sup>+</sup> + 8e<sup>-</sup> -> CH<sub>4</sub> +2 H<sub>2</sub>O (by | Methane production: CO<sub>2</sub> + 8 H<sup>+</sup> + 8e<sup>-</sup> -> CH<sub>4</sub> +2 H<sub>2</sub>O (by methanogens) | ||
====Eh==== | ====Eh==== | ||
During the | During the succession of anaerobic oxidation processes, the redox potential (Eh) of flooded soils will decrease as a result of the reduced products formed. Approximate values for redox potentials associated with specific oxidation-reduction process are as follows: | ||
{| width="300" border="1" | {| width="300" border="1" | ||
|----- bgcolor ="orange" | |||
| width="200" height="23" | Observation | | width="200" height="23" | Observation | ||
| width="84" | Eh (mV) | | width="84" | Eh (mV) | ||
|- | |- | ||
| Disappearance of oxygen | | Disappearance of oxygen | ||
| Line 63: | Line 76: | ||
| +200 | | +200 | ||
|- | |- | ||
| Appearance of ferrous ions | | Appearance of ferrous iron ions | ||
| +120 | | +120 | ||
|- | |- | ||
| Line 73: | Line 86: | ||
|} | |} | ||
===Solubility/ | ===Solubility/Mobility of Minerals=== | ||
Since the toxicity, solubility, mobility,and bioavailability | Since the toxicity, solubility, mobility, and bioavailability for a given element or compound is mainly influenced by soil solution reduction potential and pH, flooded soil conditions play an important role in the mobility of trace metal, nutrients, and minerals. | ||
What over watering looks like in a common house plant | ====Plant Nutrient Availability==== | ||
[[Image:overwater.jpg|left|frame|What over-watering looks like in a common house plant]]Flooded soils can prevent efficient gas exchange between the plant root and the soil. pH plays a major role in healthy plant growth. In flooded soils, under anaerobic conditions, the pH will tend to rise initially. Denitrification of soil nitrate to nitrogen gas plays a major role in the rise of pH levels. Flooding results in poor soil aeration because the supply of oxygen to flooded soil is severely limited. Oxygen deficiency is likely the most important environmental factor that triggers growth inhibition and injury in flooded plants. Microorganisms will begin to use available plants nutrients as alternative electron acceptors, such as sulfate, nitrate and iron(III). | |||
Experiments have been done on soybean plants to show the effects of flooded soils. Flood duration effects on soybean plants resulted in yellowing and abscission of leaves at the lower nodes, stunting, and reduced dry weight and seed yield. Canopy height and dry weight decreased linearly with duration of the flood at both growth stages. Growth rates were 25 to 35% less when soybeans were flooded (3). | |||
==Key Microbial Processes and Organisms Involved== | |||
==Key | |||
===Microbial processes=== | ===Microbial processes=== | ||
==== | ====Microbial Activity==== | ||
In anaerobic respiration oxygen is replaced by other compounds as TEA (terminal electron acceptors). Some important terminal electron acceptors include iron, nitrate, sulfate, and manganese. These processes | In anaerobic respiration oxygen is replaced by other compounds as [http://en.wikipedia.org/wiki/Electron_acceptor TEA (terminal electron acceptors)]. Some important terminal electron acceptors include iron, nitrate, sulfate, and manganese. These processes are primarily driven by microbial activity. Energy yields of alternative electron acceptors are lower than that of aerobic respiration, in which oxygen is utilized as a TEA (see [[#Electron Tower]] theory. As available oxygen declines, organisms that thrive under anoxic conditions proliferate using alternative electron acceptors. The order in which available electron acceptors are consumed can generally be predicted by the electron tower and associated energy yields of electron pairs. Changes in redox conditions of flooded soils over time reflects the successive availability of TEA's from the electron tower, and will govern which microbes will thrive, as those able to use these available alternative electron acceptors. Flooding also alters microbial flora in soil by decreasing the O<sub>2</sub> concentration. Fermentation is a major biochemical processes responsible for organic matter decomposition in flooded soils. Eh levels can affect which compounds are fermented. These levels will tend to gradually drop in flooded soils. | ||
==== | ====Fermentation under Anoxic Conditions==== | ||
[[Image:anaerobic decomposition.png|thumb|left|300px|Organic matter decomposition pathways . [[Richardson and Vepraskas]]]] | [[Image:anaerobic decomposition.png|thumb|left|300px|Organic matter decomposition pathways . [[Richardson and Vepraskas]]]] | ||
There are many types of fermentative bacteria, such as the genus ''[[Bacillus]]'', ''[[Clostridium]]'', and ''[[Lactobacillus]]'' | There are many types of fermentative bacteria in soils, such as the genus ''[[Bacillus]]'', ''[[Clostridium]]'', and ''[[Lactobacillus]]''. 4 ATP molecules per molecule of glucose are produced by fermentation, while 38 ATP molecules are produced by aerobic respiration. Although the energy yield via fermentation is less than respiration, fermentation plays an important role in anaerobic respiration for obligate and facultative anaerobic bacteria, including denitrifier, Fe<sup>3+</sup>, Mn<sup>4+</sup>, SO<sub>4</sub><sup>2-</sup>, reducers, and methanogens. Sugar (glucose or fructose) is broken down into simple compounds (e.g. formate, acetate, and ethanol) during fermentation. Also, numerous fermentation products, such as carbon dioxide, fatty acid, lactic, alcohols, are released into soils. These compounds serve as substrates for other anaerobic bacteria. Thus, low molecular weight organic compounds produced from fermentation influence the reduction of Fe(III), Mn(IV), SO<sub>4</sub><sup>2-</sup>, and CO<sub>2</sub>(Richardson And Vepraskas 2000). | ||
---- | ---- | ||
===Organisms involved in Flooded Soils=== | |||
====Nitrate Reducing Bacteria==== | |||
When available oxygen is depleted and nitrate is available, denitrification, the reduction of NO<sub>3</sub><sup>-</sup> to NO,N<sub>2</sub>O,or N<sub>2</sub>, primarily occurs. | |||
Denitrification is carried out by obligate respiratory bacteria belonging to the genera ''[[Agrobacterium]]'', ''[[Alcaligenes]]'', ''[[Bacillus]]'', ''[[Paracoccus denitrificans]]'', ''[[Pseudomonas]]'' and ''[[Thiobacillus]]'' (Knowles, 1982). Nitrate ammonification found in facultative anaerobic bacteria belonging to the genera ''[[Bacillus]]'', ''Citrobacter'' and ''[[Aeromonas]]'', or members of the ''Enterobacteriaceae'' (Cole and Brown, 1980; Smith and Zimmerman, 1981; MacFarlane and Herbert, 1982). Strictly anaerobic bacteria belonging to the genus ''[[Clostridium]]'' are also able to reduce nitrate to ammonia (Hasan and Hall, 1975). | |||
Pure culture studies show evidence that nitrate reduction may also occur in the presence of oxygen (Kuenen and Robertson, 1987). | |||
====Iron/Manganese Reducing Bacteria==== | |||
====Iron/Manganese | |||
Most microorganisms can reduce Mn<sup>4+</sup> and Fe <sup>3+</sup>. | Most microorganisms can reduce Mn<sup>4+</sup> and Fe <sup>3+</sup>. | ||
Ferrous iron is used as electron acceptor by iron-reducing bacteria such as ''[[Geobacter]]([[Geobacter metallireducens]] and [[Geobacter sulfurreducens]]),Shewanella putrefaciens,[[Desulfovibrio]], [[Pseudomonas]],'' and ''[[Thiobacillus]]''(Lovley 1993). ''[[Bacillus]], [[Geobacter]],'' and ''[[Pseudomonas]]'' are representative manganese-reducing bacteria. | Ferrous iron is used as electron acceptor by iron-reducing bacteria such as ''[[Geobacter]]([[Geobacter metallireducens]] and [[Geobacter sulfurreducens]]),Shewanella putrefaciens,[[Desulfovibrio]], [[Pseudomonas]],'' and ''[[Thiobacillus]]''(Lovley 1993). ''[[Bacillus]], [[Geobacter]],'' and ''[[Pseudomonas]]'' are representative manganese-reducing bacteria. | ||
Different forms of ferric oxides exist in aerobic | Different forms of ferric iron oxides exist in drained aerobic soils as well as in waterlogged soils. Not all forms of ferric oxides are equally suitable for reduction by ferric oxide reducer bacteria (Gotoh and Patrick, 1974; Schwertmann and Taylor, 1977). In general, amorphous forms are more efficient for ferric reducer bacteria than crystalline forms (Lovely and Phillips, 1986). The reduction of ferric oxide may release phosphate and trace elements that are adsorbed to amorphous ferric oxide and thus enhance availability of these compounds in the soil (Lovely and Phillips, 1986). | ||
====Sulfate Reducing Bacteria==== | |||
Bacteria can use organic compounds as an electron donor and sulfate as an electron acceptor. This reaction for acetate as electron donor is as follows: | |||
CH<sub>3</sub>COO<sup>-</sup> + SO<sub>4</sub><sup>2-</sup> + 3 H<sup>+</sup> ---> 2CO<sub>2</sub> + H<sub>2</sub>S + 2 H<sub>2</sub>O | |||
This reaction is carried out by sulfate-reducing bacteria such as ''[[Desulfobacter]]'', ''Desulfobulbus'', ''[[Desulfococcus]]'', ''[[Desulfovibrio]]'', ''[[Desulfosarcina]]'',''Desulfotomaculum'',and ''Desulfonema''(Langston and Bebiano 1998, Sylvia 2004). Some of the sulfate reducing bacteria oxidizes the organic compounds completely to CO2 and some other stop after producing acetate as an intermediate of oxidation. Hydrogen sulfide gas produced via anaerobic respiration causes the rotten egg odor. | |||
Recently, Boetius et al and Orphan et al revealed that a consortium of ''[[Desulfosarcina]]'' and methanotrophic archaea in the sediments contribute to control methane gas. | |||
CH<sub>4</sub> +SO<sub>4</sub> <sup>2</sup>------->HCO<sub>3</sub>+HS <sup>-</sup>+H<sub>2</sub>O | |||
====[[Methanogens]]==== | ====[[Methanogens]]==== | ||
Methanogen products less energy than other | Methanogen products less energy than other reducing reaction because the reduction of carbon dioxide occur under the most anaerobic and reduced conditions(see [[#Electron tower]] section). Thus, the activity of methanogen is repressed until other alternative terminal electron acceptor such as Fe(III), NO<sub>3</sub><sup>-</sup>,and SO<sub>4</sub><sup>2-</sup>, have been depleted. | ||
Methanogen (e.g ''Methanobacterium formicum'', ''Methanobacterium bryantii'', ''Methanobacterium thermo-autrotrophicum'', and etc ) can use CO<sub>2</sub> and produce methane (Langston and Bebiano 1998) | Methanogen (e.g ''Methanobacterium formicum'', ''Methanobacterium bryantii'', ''Methanobacterium thermo-autrotrophicum'', and etc ) can use CO<sub>2</sub> and produce methane (Langston and Bebiano 1998) | ||
| Line 132: | Line 134: | ||
==Greenhouse Gas Emissions from Flooded Soils== | ==Greenhouse Gas Emissions from Flooded Soils== | ||
Flooded soils are dynamic ecosystems that play an important role in biogeochemical cycling and in the production of greenhouse gases. Methane (CH<sub>4</sub><sup>+</sup>) and nitrous oxide (N<sub>2</sub>O) are produced as byproducts of anaerobic metabolism in the low-redox zones characteristic of flooded soils, where oxygen is lacking. Carbon dioxide (CO<sub>2</sub>), which receives widespread attention as a greenhouse gas and potential source of global warming, may also be produced at the interface of anaerobic-aerobic zones through the consumption of methane gas. However, it should be noted that from a global standpoint methane and nitrous oxide on a per molecule basis have the potential to contribute 25x and 300x more to global warming over the next century than carbon dioxide, respectively (Schlesinger, 1997). Thus the conversion of methane gas to carbon dioxide essentially reduces the greenhouse gas effect by 25x per molecule per 100 years. According to Matthews and Fung (1987), an estimated 3.6% of terrestrial land is classified as wetlands, and although this number continues to decline (Schlesinger, 1997) the effect of flooded soils to the global climate is clear. | Flooded soils are dynamic ecosystems that play an important role in biogeochemical cycling and in the production of greenhouse gases. Methane (CH<sub>4</sub><sup>+</sup>) and nitrous oxide (N<sub>2</sub>O) are produced as byproducts of anaerobic metabolism in the low-redox zones characteristic of flooded soils, where oxygen is lacking. Carbon dioxide (CO<sub>2</sub>), which receives widespread attention as a greenhouse gas and potential source of global warming, may also be produced at the interface of anaerobic-aerobic zones through the consumption of methane gas. However, it should be noted that from a global standpoint methane and nitrous oxide on a per molecule basis have the potential to contribute 25x and 300x more to global warming over the next century than carbon dioxide, respectively (Schlesinger, 1997). Thus the conversion of methane gas to carbon dioxide essentially reduces the greenhouse gas effect by 25x per molecule per 100 years. According to Matthews and Fung (1987), an estimated 3.6% of terrestrial land is classified as wetlands, and although this number continues to decline (Schlesinger, 1997) the effect of flooded soils to the global climate is clear. | ||
===Methane | ===Methane Production; Methanogenesis=== | ||
Methane production occurs exclusively in anaerobic conditions by a group of Archaea known as methanogens. These microbes are obligatory, and require extremely low redox conditions in the range of -100mV (see electron tower theory, section 2.1.1) (Sylvia, 2005). If oxygen is introduced into the system, methanogenesis ceases; thus, the process of methanogenesis depends on saturated soil conditions. | [[Image:Methane.jpg|thumb|300px|A natural source of methane gas]] | ||
Methane production occurs exclusively in anaerobic conditions by a group of Archaea known as methanogens. These microbes are obligatory, and require extremely low redox conditions in the range of -100mV (see [[#electron tower]] theory, section 2.1.1) (Sylvia, 2005). If oxygen is introduced into the system, methanogenesis ceases; thus, the process of methanogenesis depends on saturated soil conditions. | |||
Methanogenesis can occur via one of two pathways: either by 1) CO<sub>2</sub> reduction or by 2) acetate fermentation. | Methanogenesis can occur via one of two pathways: either by 1) CO<sub>2</sub> reduction or by 2) acetate fermentation. | ||
| Line 149: | Line 152: | ||
Despite the clear effect of increasing temperatures on the rate of methanogenesis, the actual impact of global warming on methane production rates in wetlands and permafrost regions is highly unpredictable. Because methanogenesis requires anoxic conditions, any drying of flooded soil environments would both decrease methane production and increase methane oxidation, reducing overall methane emissions. Alternatively, warmer climates could increase growing seasons, which would increase methane emissions (Sylvia, 2005). | Despite the clear effect of increasing temperatures on the rate of methanogenesis, the actual impact of global warming on methane production rates in wetlands and permafrost regions is highly unpredictable. Because methanogenesis requires anoxic conditions, any drying of flooded soil environments would both decrease methane production and increase methane oxidation, reducing overall methane emissions. Alternatively, warmer climates could increase growing seasons, which would increase methane emissions (Sylvia, 2005). | ||
===CO<sub>2</sub> | ===CO<sub>2</sub> Production via Methane Consumption: Methanotrophy=== | ||
Some of the methane produced via methanogenesis in flooded soils may be consumed and oxidized to CO<sub>2</sub> at the interface of the anaerobic-aerobic zones. This process occurs primarily by a group of bacteria known as methanotrophs. These microbes can be found in surface layers of wetland soils and unsaturated upland soils, and may be exposed to very high concentrations of methane gas, sometimes amounting to 10% or more of the dissolved gases. Methane is thought to be the only source of C and energy for these bacteria. | Some of the methane produced via methanogenesis in flooded soils may be consumed and oxidized to CO<sub>2</sub> at the interface of the anaerobic-aerobic zones. This process occurs primarily by a group of bacteria known as methanotrophs. These microbes can be found in surface layers of wetland soils and unsaturated upland soils, and may be exposed to very high concentrations of methane gas, sometimes amounting to 10% or more of the dissolved gases. Methane is thought to be the only source of C and energy for these bacteria. | ||
| Line 160: | Line 163: | ||
Alternatively, if nitrogen is extremely limiting the addition of nitrogen will stimulate methanotrophy and actually increase methane consumption. So although it is generally expected that adding N-fertilizer will decrease CH<sub>4</sub><sup>+</sup> consumption and lead to increased global warming potential, sometime the opposite effect may occur. (Sylvia, 2005). | Alternatively, if nitrogen is extremely limiting the addition of nitrogen will stimulate methanotrophy and actually increase methane consumption. So although it is generally expected that adding N-fertilizer will decrease CH<sub>4</sub><sup>+</sup> consumption and lead to increased global warming potential, sometime the opposite effect may occur. (Sylvia, 2005). | ||
===Nitrous | ===Nitrous Oxide; Denitrification=== | ||
Denitrification is an anaerobic process in which nitrate serves as the terminal electron acceptor, and generally some source of organic carbon is the electron donor (also | Denitrification is an anaerobic process in which nitrate serves as the terminal electron acceptor, and generally some source of organic carbon is the electron donor (also H<sub>2</sub> may serve as a donor). | ||
In this process, nitrate is oxidized to nitric oxide, then nitrous oxide, and then fully oxidized to dinitrogen: | In this process, nitrate is oxidized to nitric oxide, then nitrous oxide, and then fully oxidized to dinitrogen: | ||
| Line 174: | Line 177: | ||
Factors affecting nitrous oxide production include oxygen, pH, and the ratio of nitrate to available C. Although denitrification rates decrease with increasing oxygen, the proportion of N evolved as nitrous oxide actually increases with increasing oxygen. Low pH generally inhibits the reduction of N<sub>2</sub>O to N<sub>2</sub>; thus at low pH, N<sub>2</sub>O will likely dominate. However, highly acidic soils have low N availability and low nitrification and denitrification rates. Thus, the highest rate of nitrous oxide production from denitrification occurs in moist soils that cycle N rapidly (Sylvia, 2005). | Factors affecting nitrous oxide production include oxygen, pH, and the ratio of nitrate to available C. Although denitrification rates decrease with increasing oxygen, the proportion of N evolved as nitrous oxide actually increases with increasing oxygen. Low pH generally inhibits the reduction of N<sub>2</sub>O to N<sub>2</sub>; thus at low pH, N<sub>2</sub>O will likely dominate. However, highly acidic soils have low N availability and low nitrification and denitrification rates. Thus, the highest rate of nitrous oxide production from denitrification occurs in moist soils that cycle N rapidly (Sylvia, 2005). | ||
==Current Research== | |||
Current research topics on the issue of flooded soils are heavily focused on greenhouse gas emissions produced as a result of the low redox conditions characteristic of these ecosystems. Other research topics may address impacts to plant growth, and chemical, physical, and biological aspects of flooded soils. Following is a list of recent journal review articles focused on a range of current research topics related to flooded soil environments for the interested reader: | |||
1. Shangping Xu, Peter R. Jaffe and Denise L. Mauzerall, A process-based model for methane emission from flooded rice paddy systems, Ecological ModellingVolume 205, Issues 3-4, , 24 July 2007, Pages 475-491. | |||
(http://www.sciencedirect.com/science/article/B6VBS-4NHV759-1/2/3126b5403a44c51c5d8d4160382d848e) | |||
2. HANK GREENWAY , WILLIAM ARMSTRONG , and TIMOTHY D. COLMER | |||
Conditions Leading to High CO2 (>5 kPa) in Waterlogged–Flooded Soils and Possible Effects on Root Growth and Metabolism | |||
AOBPreview published on July 1, 2006, DOI 10.1093/aob/mcl076. | |||
Ann Bot 98: 9-32. | |||
3. Kazunori Minamikawa and Naoki Sakai, The effect of water management based on soil redox potential on methane emission from two kinds of paddy soils in Japan, Agriculture, Ecosystems & EnvironmentVolume 107, Issue 4, , 30 May 2005, Pages 397-407. | |||
(http://www.sciencedirect.com/science/article/B6T3Y-4FY9M5H-1/2/98a4074277436af97833c16a823b326c) | |||
Keywords: Methane; Water management; Soil redox potential; Oryza sativa L.; Rice yield | |||
4. Makoto Kimura, Jun Murase and Yahai Lu, Carbon cycling in rice field ecosystems in the context of input, decomposition and translocation of organic materials and the fates of their end products (CO2 and CH4), Soil Biology and BiochemistryVolume 36, Issue 9, , September 2004, Pages 1399-1416. | |||
(http://www.sciencedirect.com/science/article/B6TC7-4C9G7KP-4/2/ceb77d1803f5d02e51bb773f3163cdcb) | |||
5. Sahrawat KL, Fertility and organic matter in submerged rice soils, Current Science. Volume 88, Issues 3-4, 2005, Pages 735-739. | |||
6. Conrad R, Microbial ecology of methanogens and methanotrophs, Advances in Agronomy. Volume 96, 2007, Pages 1-63. | |||
7. Ralf Conrad, Christoph Erkel and Werner Liesack, Rice Cluster I methanogens, an important group of Archaea producing greenhouse gas in soil, Current Opinion in BiotechnologyVolume 17, Issue 3, , Environmental biotechnology/Energy biotechnology, June 2006, Pages 262-267. | |||
(http://www.sciencedirect.com/science/article/B6VRV-4JRVFV8-1/2/babb823ea30e51d7445d6861d7d334aa) | |||
==References== | ==References== | ||
(1) Lecture 5 of Kate Scow. 2008. Microbial Metabolism. Unpublished, University of California, Davis. | |||
( | (2) Schlesinger, W.H. 1997. Biogeochemistry: An Analysis of Global Change. 2nd ed. Elsevier Academic Press, Amsterdam. | ||
(3) [http://books.google.com/books?id=1l4GAAAACAAJ&dq=soil+microbiology+sylvia&ei=thzbR9j6Hpu8swPmxOXzAQ Silvia, D.M., et al. 2005. Principles and Applications of Soil Microbiology. 2nd ed. Pearson Prentice Hall, New Jersey.] | |||
(4) [http://books.google.com/books?id=v6cGPMRmPYYC&pg=PA940&dq=J.+Kotz,+P.+Treichel,+G.+Weaver.+2006.+Chemistry+and+Chemical+Reactivity&ei=Bx3bR5yFF5-OtgOdqe3sAQ&sig=2IiJ_hdME4k5k06zd-H35y1cxC8#PPP1,M1 J. Kotz, P. Treichel, G. Weaver. 2006. Chemistry and Chemical Reactivity sixth edition.] | |||
( | (5)[http://books.google.com/books?id=xEFIEOjfxx0C&printsec=frontcover&dq=Wetland+richardson&ei=7xvbR4vMH4jysgOg6vnpAQ&sig=bDFNpm0lUibISaIaERoMAu5-K34 J.L Richardson and M.J Vepraskas., 2000 Wetland soils: Genesis, Hydrology, Landscapes, and Classification., CRC press LLC.] | ||
( | (6) Flood Duration Effects on Soybean Growth and Yield | ||
http://agron.scijournals.org/cgi/content/abstract/81/4/631 | http://agron.scijournals.org/cgi/content/abstract/81/4/631 | ||
( | (7)[http://books.google.com/books?id=pr7kAQAACAAJ&dq=Advances+in+Agricultural+Microbiology,&ei=ddHdR6qDJYG-sgPeyMXyAQ Knowles, R. 1982. Denitrification in Soils, pp. 246-266, In: N.S. Subba Rao (ed.) Advances in Agricultural Microbiology, Butterworth Sci. Pub., London, UK] | ||
(8) [http://www.blackwell-synergy.com/doi/abs/10.1111/j.1574-6941.1980.tb01578.x Cole JA, Brown CM (1980) Nitrite reduction to ammonium by fermentative bacteria: a short circuit in the biological nitrogen cycle. FEMS Microbiol Lett 7: 65–72] | |||
(5) Matthews, E. and I. Fung. 1987. Methane Emission from Natural Wetlands: Global Distribution, Area, and Environmental Characteristics of Sources. Global Biogeochemical Cycles 1: 61-86. | (9) [http://soil.scijournals.org/cgi/content/abstract/45/5/865 Smith SM, Zimmerman K (1981) Nitrous oxide production by nondenitrifying soil nitrate reducers. Soil Sci Soc Am J 45:865–871] | ||
(10) MacFarlane, GT and Herbert, RA.,(1982) Nitrate dissimilation by Vibrio spp. isolated from estuarine sediments., Microbiology [J. GEN. MICROBIOL.]. Vol. 128, no. 10, pp. 2379-2383. | |||
(11) S. M. Hasan and J. B. Hall, The physiological function of nitrate reduction inClostridium perfringens. J. Gen. Microbiol. 87 (1975), pp. 120–128 | |||
(12) Kuenen, J.G. and Robertson, L.A., 1988. Ecology of nitrification and denitrification. In: Cole, J.A. and Ferguson, S.J., Editors, 1988. The Nitrogen and Sulphur Cycles. 42nd Symposium of The Society for General Microbiology, University of Southampton, Cambridge University Press, Cambridge, pp. 161–218. | |||
(13)[http://arjournals.annualreviews.org/doi/abs/10.1146/annurev.mi.47.100193.001403?journalCode=micro Derek R. Lovley., 1993., Dissimilatory metal reduction. Annu Rev Microbiol. Vol 47 pp: 263-90.] | |||
(14) Gotoh, S and Patrick, W HJr 1974 Transformations of iron in waterlogged soils as influenced by redox potential and pH. Soil Sci. Soc. Am. Proc. 38, 66–71. | |||
(15) [Schwertmann U & Taylor RM (1977) Iron oxides. Chap 5 In: Dixon JB & Weed SB (Eds) Minerals in Soil Environments (pp 145–180). Soil Sci. Soc. AM:; Madison, WI] | |||
(18) [http://aem.asm.org/cgi/reprint/52/4/751 Derek R. Lovley and Elizabeth J.P Phillips., Availability of Ferric Iron for Microbial Reduction in Bottom Sediments of the Freshwater Tidal Potomac River., Appl Environ Microbiology. 1986, p. 751-757] | |||
(19) [http://books.google.com/books?id=0GAvKQJ2JuwC&printsec=frontcover&vq=Wenk+H.R+and+Bulakh+A+.,+2004:+Minerals.+Their+constitution+and+origin.+Cambridge+University+Press&source=gbs_summary_r Wenk H.R and Bulakh A ., 2004: Minerals. Their constitution and origin. Cambridge University Press.] | |||
(20) [http://books.google.com/books?id=9K5I0ZPQd54C&pg=PA219&lpg=PA219&dq=%22LANGSTON%22+%228+Metal+handling%22&source=web&ots=qVFErWWjbn&sig=Iyh9_HN4S6PZU4jdXrMuWbLqcpI&hl=en#PPR11,M1 WJ Langston, MJ Bebianno, ,and GR Burt, ., (1998) "Metal handling strategies in molluscs" In: Langston, WJ, Bebiano, MJ eds. , Metal metabolism in the aquatic environment, Chapman and Hall, London, United Kingdom, pp 219-272] | |||
(21) Matthews, E. and I. Fung. 1987. Methane Emission from Natural Wetlands: Global Distribution, Area, and Environmental Characteristics of Sources. Global Biogeochemical Cycles 1: 61-86. | |||
(22) [http://books.google.com/books?id=zHIhWM5R9LcC&printsec=frontcover&dq=Y.Chen+and+Y+Avnimelech&source=gbs_summary_r#PPA118,M1 Y. Chen and Y . Avnimelech., (1986)., The Role of Organic Matter in Modern Agriculture., Springer., Developments in Plant and Soil Sciences , Vol. 25., p118] | |||
See [[#Current Research]] section for a list of journals for additional information on the topic of flooded soils. | |||
Edited by students of [mailto:kmscow@ucdavis.edu Kate Scow] | Edited by students of [mailto:kmscow@ucdavis.edu Kate Scow] | ||
<!-- Do not edit or remove this line --> [[Category:Pages edited by students of Kate Scow at UC Davis]] | |||
Latest revision as of 05:41, 21 January 2016
Introduction
Flooded soils occur with complete water saturation of soil pores, and generally result in anoxic conditions of the soil environment. Flooded soil environments may include such ecosystem as: rice paddies; wetlands (swamps, marshes, and bogs); compacted soils; and post-rain soils (Scow, 2008). Additionally, similar redox conditions (where oxygen is lacking) can also be found within soil aggregates and along pollutant plumes, and thus many of the concepts discussed in this section may be applied to those environments.
Oxygen is only sparingly soluble in water and diffuses much more slowly through water than through air (Schlesinger, 1997). What little oxygen that is present in saturated soils in the form of dissolved O2 is quickly consumed through metabolic processes. Oxygen is used as terminal electron acceptor via respiration by roots, soil microbes, and soil organisms (Sylvia, 2005), and is lost from the soil system in the form of carbon dioxide (CO2). Heterotrophic respiration may completely deplete oxygen in flooded soils; and these effects may be observed within only a few millimeters of the soil surface (Schlesinger, 1997).
Due to the deficiency of oxygen in flooded soils, those organisms inhabiting flooded soils must be able to survive with little to no oxygen. Although energy yields are much greater with oxygen than with any other terminal electron acceptor (see #Electron tower theory, section 2.1.1), under anoxic conditions anaerobic and facultative microbes can use alternative electron acceptors such as nitrate, ferric iron (Fe III), manganese (IV) oxide, sulfate, and carbon dioxide to produce energy and build biomass.
Microbial transformations of elements in anaerobic soils play a large role in biogeochemical cycling of nutrients and in greenhouse gas emissions. Changes in the oxidation state of terminal electron acceptors may result in nutrient loss from the system via volatilization or leaching. Anaerobic microbial processes including denitrification, methanogenesis, and methanotrophy are responsible for releasing greenhouse gases (N2O, CH4, CO2) into the atmosphere (Schlesinger, 1997).
Processes


In general, flooded soils occur due to seasonal flooding or agricultural activity. Flooded soils can be often converted into non-flooded soils by the water level fluctuation and drainage. Through this variation of soil conditions, various gases are emitted into the atmosphere and environmental factors, such as redox potential (Eh), pH, acidity, alkalinity, and salinity, are continuously changing. As explained in the #Introduction, microorganisms can use alternative terminal electron acceptors (such as nitrate, perchlorate, sulfate, and carbon dioxide) when dissolved oxygen is absent. Microbes will successively use electron acceptors according to the order of energy yields resulting from electron acceptor utilization indicted on the electron tower (see #Electron Tower theory). The progression of electron acceptor utilization may also be observed in soil aggregates and pollutant plumes.
Oxidation/Reduction (Redox) Reaction
In redox reactions, one molecule (the reducing agent) loses electrons and another molecule (the oxidizing agent) accepts electrons. A classic example well known in the process of cellular respiration is when glucose (the reducing agent) reacts with oxygen (the oxidizing agent)and is oxidized to carbon dioxide. In this reaction, oxygen is reduced to water. Oxygen is the most common and highest energy yielding electron acceptor, and some organisms (strict aerobes) can not live long without it.(6) In flooded soils oxygen is typically not available. Facultative and strict anaerobic bacteria have the ability to use other oxidizing agents/electron acceptors to carry out respiration. Anaerobic and facultative bacteria will use the electron acceptor which yields the highest energy, or the acceptor which is most readily available. The availability and concentration of electron acceptors changes as the soil profile increases in depth.
Electron Tower
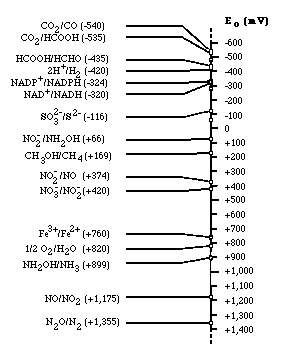
Electron tower theory explains the utilization order of electron acceptor for respiration. Depending on the type of electron acceptors used by microorganisms, microbes can be classified as strict aerobes, obligate anaerobes, and facultative anaerobes. Strict aerobes can not live under anoxic condition; on the contrary, obligate anaerobes can never use oxygen as electron acceptor. However, facultative anaerobes can live in both aerobic and anaerobic condition. If oxygen is plentiful, they tend to use oxygen because microorganisms gain much energy from reducing oxygen rather than other electron acceptors. When there is no more available oxygen in solution, microbes will choose to use nitrate as an electron acceptor (if available). Thus, obligate anaerobes and facultative anaerobes use alternative electron acceptors in the order of electron acceptor having the most reducing energy. Oxygen is the most efficient electron acceptor, while carbon dioxide has the least amount of reduction potential.
Gleyed Soils and Recovery to Aerobic Conditions
Soil Gleying: Gleying is a phenomenon in which waterlogged soils are discolored by accumulation of Fe(II) due to reduction of ferric iron into ferrous iron (Lovely 1991). Although ferric iron exists as an insoluble form in flooded soils, more ferrous iron can accumulate by the reduction of ferric iron over time. This results in a greenish, blue, grey soil color. In general Fe(III)-reducing fermentative bacteria can be readily isolated from gleyed soils. The black color of soils/solution is frequently observed in flooded soil. This may result from the formation of iron sulfides (FeS) and pyrite (FeS2) (Wenk and Bulakh 2004).
Recovery to Aerobic Conditions When waterlogged soils drain, the Eh starts to increase as oxygen diffuses into soil pores. Plentiful oxygen represses the activity of anaerobes, which results in an increase of aerobic microbes. If oxygen diffuses deep into the soil profile, the production of H2S ceases. Under aerobic conditions, ferrous iron is oxidized by iron-oxidizing bacteria, resulting in the formation of ferric oxides or ferric hydroxide minerals. The gray color in soil changes to a red, yellow, or brown color as these minerals are oxidized. At higher Eh zones ( > 500 mV), undecomposed soil organic matter is used as an electron donor by aerobes and converted to water and CO2 (Richardson and Vepraskas 2000). In aerobic condition, manganese can be readily oxidized to MnO2. Black soil is often shown in aerated soil because manganese oxide is a mineral with a black color.
Variation of pH and Eh
pH
Neutral pH soil
When soil is saturated with water, pH drops at first due to organic acid produced from fermentation. Then, pH gradually starts to rise as a result of the buffering capacity of the soil. The half reactions of hydrogen consumption are as follow;
Aerobic respiration: ½ O2 + 2e- + 2H+ -> H2O (by facultative anaerobes and aerobes)
Denitrification: 2NO3- + 12 H+ +10e- -> N2+6H2O (by denitrifiers)
Manganese reduction: MnO2 + 4H+ + 2e- ->Mn2+ + 2H2O (by manganese reducing bacteria)
Iron reduction: Fe(OH)3 + 3 H+ + 2e- -> Fe2+ + 2H2O (by iron reducing bacteria)
Sulfate reduction: SO42- + 10H+ +8e- -> H2S + 4H2O (by sulfate reducing bacteria)
Methane production: CO2 + 8 H+ + 8e- -> CH4 +2 H2O (by methanogens)
Eh
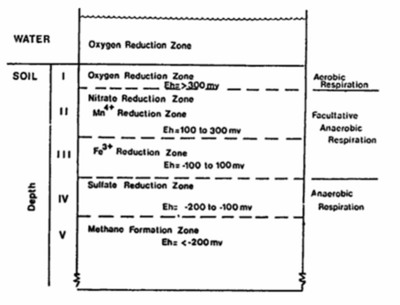
During the succession of anaerobic oxidation processes, the redox potential (Eh) of flooded soils will decrease as a result of the reduced products formed. Approximate values for redox potentials associated with specific oxidation-reduction process are as follows:
| Observation | Eh (mV) |
| Disappearance of oxygen | +330 |
| Disappearance of nitrate | +220 |
| Appearance of manganese ions | +200 |
| Appearance of ferrous iron ions | +120 |
| Disappearance of sulfate | -150 |
| Appearance of methane | -250 |
Solubility/Mobility of Minerals
Since the toxicity, solubility, mobility, and bioavailability for a given element or compound is mainly influenced by soil solution reduction potential and pH, flooded soil conditions play an important role in the mobility of trace metal, nutrients, and minerals.
Plant Nutrient Availability
Flooded soils can prevent efficient gas exchange between the plant root and the soil. pH plays a major role in healthy plant growth. In flooded soils, under anaerobic conditions, the pH will tend to rise initially. Denitrification of soil nitrate to nitrogen gas plays a major role in the rise of pH levels. Flooding results in poor soil aeration because the supply of oxygen to flooded soil is severely limited. Oxygen deficiency is likely the most important environmental factor that triggers growth inhibition and injury in flooded plants. Microorganisms will begin to use available plants nutrients as alternative electron acceptors, such as sulfate, nitrate and iron(III).
Experiments have been done on soybean plants to show the effects of flooded soils. Flood duration effects on soybean plants resulted in yellowing and abscission of leaves at the lower nodes, stunting, and reduced dry weight and seed yield. Canopy height and dry weight decreased linearly with duration of the flood at both growth stages. Growth rates were 25 to 35% less when soybeans were flooded (3).
Key Microbial Processes and Organisms Involved
Microbial processes
Microbial Activity
In anaerobic respiration oxygen is replaced by other compounds as TEA (terminal electron acceptors). Some important terminal electron acceptors include iron, nitrate, sulfate, and manganese. These processes are primarily driven by microbial activity. Energy yields of alternative electron acceptors are lower than that of aerobic respiration, in which oxygen is utilized as a TEA (see #Electron Tower theory. As available oxygen declines, organisms that thrive under anoxic conditions proliferate using alternative electron acceptors. The order in which available electron acceptors are consumed can generally be predicted by the electron tower and associated energy yields of electron pairs. Changes in redox conditions of flooded soils over time reflects the successive availability of TEA's from the electron tower, and will govern which microbes will thrive, as those able to use these available alternative electron acceptors. Flooding also alters microbial flora in soil by decreasing the O2 concentration. Fermentation is a major biochemical processes responsible for organic matter decomposition in flooded soils. Eh levels can affect which compounds are fermented. These levels will tend to gradually drop in flooded soils.
Fermentation under Anoxic Conditions
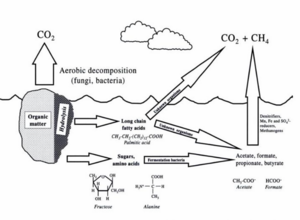
There are many types of fermentative bacteria in soils, such as the genus Bacillus, Clostridium, and Lactobacillus. 4 ATP molecules per molecule of glucose are produced by fermentation, while 38 ATP molecules are produced by aerobic respiration. Although the energy yield via fermentation is less than respiration, fermentation plays an important role in anaerobic respiration for obligate and facultative anaerobic bacteria, including denitrifier, Fe3+, Mn4+, SO42-, reducers, and methanogens. Sugar (glucose or fructose) is broken down into simple compounds (e.g. formate, acetate, and ethanol) during fermentation. Also, numerous fermentation products, such as carbon dioxide, fatty acid, lactic, alcohols, are released into soils. These compounds serve as substrates for other anaerobic bacteria. Thus, low molecular weight organic compounds produced from fermentation influence the reduction of Fe(III), Mn(IV), SO42-, and CO2(Richardson And Vepraskas 2000).
Organisms involved in Flooded Soils
Nitrate Reducing Bacteria
When available oxygen is depleted and nitrate is available, denitrification, the reduction of NO3- to NO,N2O,or N2, primarily occurs. Denitrification is carried out by obligate respiratory bacteria belonging to the genera Agrobacterium, Alcaligenes, Bacillus, Paracoccus denitrificans, Pseudomonas and Thiobacillus (Knowles, 1982). Nitrate ammonification found in facultative anaerobic bacteria belonging to the genera Bacillus, Citrobacter and Aeromonas, or members of the Enterobacteriaceae (Cole and Brown, 1980; Smith and Zimmerman, 1981; MacFarlane and Herbert, 1982). Strictly anaerobic bacteria belonging to the genus Clostridium are also able to reduce nitrate to ammonia (Hasan and Hall, 1975). Pure culture studies show evidence that nitrate reduction may also occur in the presence of oxygen (Kuenen and Robertson, 1987).
Iron/Manganese Reducing Bacteria
Most microorganisms can reduce Mn4+ and Fe 3+. Ferrous iron is used as electron acceptor by iron-reducing bacteria such as Geobacter(Geobacter metallireducens and Geobacter sulfurreducens),Shewanella putrefaciens,Desulfovibrio, Pseudomonas, and Thiobacillus(Lovley 1993). Bacillus, Geobacter, and Pseudomonas are representative manganese-reducing bacteria. Different forms of ferric iron oxides exist in drained aerobic soils as well as in waterlogged soils. Not all forms of ferric oxides are equally suitable for reduction by ferric oxide reducer bacteria (Gotoh and Patrick, 1974; Schwertmann and Taylor, 1977). In general, amorphous forms are more efficient for ferric reducer bacteria than crystalline forms (Lovely and Phillips, 1986). The reduction of ferric oxide may release phosphate and trace elements that are adsorbed to amorphous ferric oxide and thus enhance availability of these compounds in the soil (Lovely and Phillips, 1986).
Sulfate Reducing Bacteria
Bacteria can use organic compounds as an electron donor and sulfate as an electron acceptor. This reaction for acetate as electron donor is as follows:
CH3COO- + SO42- + 3 H+ ---> 2CO2 + H2S + 2 H2O
This reaction is carried out by sulfate-reducing bacteria such as Desulfobacter, Desulfobulbus, Desulfococcus, Desulfovibrio, Desulfosarcina,Desulfotomaculum,and Desulfonema(Langston and Bebiano 1998, Sylvia 2004). Some of the sulfate reducing bacteria oxidizes the organic compounds completely to CO2 and some other stop after producing acetate as an intermediate of oxidation. Hydrogen sulfide gas produced via anaerobic respiration causes the rotten egg odor.
Recently, Boetius et al and Orphan et al revealed that a consortium of Desulfosarcina and methanotrophic archaea in the sediments contribute to control methane gas.
CH4 +SO4 2------->HCO3+HS -+H2O
Methanogens
Methanogen products less energy than other reducing reaction because the reduction of carbon dioxide occur under the most anaerobic and reduced conditions(see #Electron tower section). Thus, the activity of methanogen is repressed until other alternative terminal electron acceptor such as Fe(III), NO3-,and SO42-, have been depleted.
Methanogen (e.g Methanobacterium formicum, Methanobacterium bryantii, Methanobacterium thermo-autrotrophicum, and etc ) can use CO2 and produce methane (Langston and Bebiano 1998)
Greenhouse Gas Emissions from Flooded Soils
Flooded soils are dynamic ecosystems that play an important role in biogeochemical cycling and in the production of greenhouse gases. Methane (CH4+) and nitrous oxide (N2O) are produced as byproducts of anaerobic metabolism in the low-redox zones characteristic of flooded soils, where oxygen is lacking. Carbon dioxide (CO2), which receives widespread attention as a greenhouse gas and potential source of global warming, may also be produced at the interface of anaerobic-aerobic zones through the consumption of methane gas. However, it should be noted that from a global standpoint methane and nitrous oxide on a per molecule basis have the potential to contribute 25x and 300x more to global warming over the next century than carbon dioxide, respectively (Schlesinger, 1997). Thus the conversion of methane gas to carbon dioxide essentially reduces the greenhouse gas effect by 25x per molecule per 100 years. According to Matthews and Fung (1987), an estimated 3.6% of terrestrial land is classified as wetlands, and although this number continues to decline (Schlesinger, 1997) the effect of flooded soils to the global climate is clear.
Methane Production; Methanogenesis
Methane production occurs exclusively in anaerobic conditions by a group of Archaea known as methanogens. These microbes are obligatory, and require extremely low redox conditions in the range of -100mV (see #electron tower theory, section 2.1.1) (Sylvia, 2005). If oxygen is introduced into the system, methanogenesis ceases; thus, the process of methanogenesis depends on saturated soil conditions.
Methanogenesis can occur via one of two pathways: either by 1) CO2 reduction or by 2) acetate fermentation.
1) CO2 + H2 --> CH4+ (CO2 reduction)
and
2) CH3COOH --> CH4+ + CO2 (acetate fermentation)
Both acetate and hydrogen are byproducts of anaerobic fermentation.
Because the process of methanogenesis is “fed” byproducts produced from a complex series of degradation processes which are themselves “fed” complex organic matter, rates of methane production are highly sensitive to changes in temperature. Methanogenesis has a Q10 value in the range of 30-40, which is substantially higher than most biochemical process (Sylvia, 2005).
Despite the clear effect of increasing temperatures on the rate of methanogenesis, the actual impact of global warming on methane production rates in wetlands and permafrost regions is highly unpredictable. Because methanogenesis requires anoxic conditions, any drying of flooded soil environments would both decrease methane production and increase methane oxidation, reducing overall methane emissions. Alternatively, warmer climates could increase growing seasons, which would increase methane emissions (Sylvia, 2005).
CO2 Production via Methane Consumption: Methanotrophy
Some of the methane produced via methanogenesis in flooded soils may be consumed and oxidized to CO2 at the interface of the anaerobic-aerobic zones. This process occurs primarily by a group of bacteria known as methanotrophs. These microbes can be found in surface layers of wetland soils and unsaturated upland soils, and may be exposed to very high concentrations of methane gas, sometimes amounting to 10% or more of the dissolved gases. Methane is thought to be the only source of C and energy for these bacteria.
Methanotrophy occurs in the following reaction:
CH4+ + 2O2 --> CO2 + 2H2O
Methane is similar in size and shape to ammonium; and there is some evidence that nitrifiers (ammonium oxidizers) can also oxidize methane (Sylvia, 1998). Because they are molecularly similar, NH4+ competes at the enzyme’s active site, inhibiting methane oxidation. As a result, methanotrophy is generally inhibited by the addition of fertilizer or excess nitrogen in the system, when ammonium levels are high.
Alternatively, if nitrogen is extremely limiting the addition of nitrogen will stimulate methanotrophy and actually increase methane consumption. So although it is generally expected that adding N-fertilizer will decrease CH4+ consumption and lead to increased global warming potential, sometime the opposite effect may occur. (Sylvia, 2005).
Nitrous Oxide; Denitrification
Denitrification is an anaerobic process in which nitrate serves as the terminal electron acceptor, and generally some source of organic carbon is the electron donor (also H2 may serve as a donor).
In this process, nitrate is oxidized to nitric oxide, then nitrous oxide, and then fully oxidized to dinitrogen:
NO2- --> NO --> N2O --> N2
However, under certain conditions the full oxidation of NO3- to N2 does not occur and nitrous oxide (N2O) is produced.
Microbes responsible include both organotrophs and lithotrophs, and this process occurs primarily by facultative anaerobes.
Although a low redox potential is important for denitrification to occur (oxygen must not be present or it will “out-compete” nitrate as a terminal electron acceptor), redox requirements are not so low that this process cannot occur within anaerobic microsites of soil aggregates.
Factors affecting nitrous oxide production include oxygen, pH, and the ratio of nitrate to available C. Although denitrification rates decrease with increasing oxygen, the proportion of N evolved as nitrous oxide actually increases with increasing oxygen. Low pH generally inhibits the reduction of N2O to N2; thus at low pH, N2O will likely dominate. However, highly acidic soils have low N availability and low nitrification and denitrification rates. Thus, the highest rate of nitrous oxide production from denitrification occurs in moist soils that cycle N rapidly (Sylvia, 2005).
Current Research
Current research topics on the issue of flooded soils are heavily focused on greenhouse gas emissions produced as a result of the low redox conditions characteristic of these ecosystems. Other research topics may address impacts to plant growth, and chemical, physical, and biological aspects of flooded soils. Following is a list of recent journal review articles focused on a range of current research topics related to flooded soil environments for the interested reader:
1. Shangping Xu, Peter R. Jaffe and Denise L. Mauzerall, A process-based model for methane emission from flooded rice paddy systems, Ecological ModellingVolume 205, Issues 3-4, , 24 July 2007, Pages 475-491. (http://www.sciencedirect.com/science/article/B6VBS-4NHV759-1/2/3126b5403a44c51c5d8d4160382d848e)
2. HANK GREENWAY , WILLIAM ARMSTRONG , and TIMOTHY D. COLMER Conditions Leading to High CO2 (>5 kPa) in Waterlogged–Flooded Soils and Possible Effects on Root Growth and Metabolism AOBPreview published on July 1, 2006, DOI 10.1093/aob/mcl076. Ann Bot 98: 9-32.
3. Kazunori Minamikawa and Naoki Sakai, The effect of water management based on soil redox potential on methane emission from two kinds of paddy soils in Japan, Agriculture, Ecosystems & EnvironmentVolume 107, Issue 4, , 30 May 2005, Pages 397-407. (http://www.sciencedirect.com/science/article/B6T3Y-4FY9M5H-1/2/98a4074277436af97833c16a823b326c) Keywords: Methane; Water management; Soil redox potential; Oryza sativa L.; Rice yield
4. Makoto Kimura, Jun Murase and Yahai Lu, Carbon cycling in rice field ecosystems in the context of input, decomposition and translocation of organic materials and the fates of their end products (CO2 and CH4), Soil Biology and BiochemistryVolume 36, Issue 9, , September 2004, Pages 1399-1416. (http://www.sciencedirect.com/science/article/B6TC7-4C9G7KP-4/2/ceb77d1803f5d02e51bb773f3163cdcb)
5. Sahrawat KL, Fertility and organic matter in submerged rice soils, Current Science. Volume 88, Issues 3-4, 2005, Pages 735-739.
6. Conrad R, Microbial ecology of methanogens and methanotrophs, Advances in Agronomy. Volume 96, 2007, Pages 1-63.
7. Ralf Conrad, Christoph Erkel and Werner Liesack, Rice Cluster I methanogens, an important group of Archaea producing greenhouse gas in soil, Current Opinion in BiotechnologyVolume 17, Issue 3, , Environmental biotechnology/Energy biotechnology, June 2006, Pages 262-267. (http://www.sciencedirect.com/science/article/B6VRV-4JRVFV8-1/2/babb823ea30e51d7445d6861d7d334aa)
References
(1) Lecture 5 of Kate Scow. 2008. Microbial Metabolism. Unpublished, University of California, Davis.
(2) Schlesinger, W.H. 1997. Biogeochemistry: An Analysis of Global Change. 2nd ed. Elsevier Academic Press, Amsterdam.
(4) J. Kotz, P. Treichel, G. Weaver. 2006. Chemistry and Chemical Reactivity sixth edition.
(6) Flood Duration Effects on Soybean Growth and Yield http://agron.scijournals.org/cgi/content/abstract/81/4/631
(10) MacFarlane, GT and Herbert, RA.,(1982) Nitrate dissimilation by Vibrio spp. isolated from estuarine sediments., Microbiology [J. GEN. MICROBIOL.]. Vol. 128, no. 10, pp. 2379-2383.
(11) S. M. Hasan and J. B. Hall, The physiological function of nitrate reduction inClostridium perfringens. J. Gen. Microbiol. 87 (1975), pp. 120–128
(12) Kuenen, J.G. and Robertson, L.A., 1988. Ecology of nitrification and denitrification. In: Cole, J.A. and Ferguson, S.J., Editors, 1988. The Nitrogen and Sulphur Cycles. 42nd Symposium of The Society for General Microbiology, University of Southampton, Cambridge University Press, Cambridge, pp. 161–218.
(13)Derek R. Lovley., 1993., Dissimilatory metal reduction. Annu Rev Microbiol. Vol 47 pp: 263-90.
(14) Gotoh, S and Patrick, W HJr 1974 Transformations of iron in waterlogged soils as influenced by redox potential and pH. Soil Sci. Soc. Am. Proc. 38, 66–71.
(15) [Schwertmann U & Taylor RM (1977) Iron oxides. Chap 5 In: Dixon JB & Weed SB (Eds) Minerals in Soil Environments (pp 145–180). Soil Sci. Soc. AM:; Madison, WI]
(19) Wenk H.R and Bulakh A ., 2004: Minerals. Their constitution and origin. Cambridge University Press.
(21) Matthews, E. and I. Fung. 1987. Methane Emission from Natural Wetlands: Global Distribution, Area, and Environmental Characteristics of Sources. Global Biogeochemical Cycles 1: 61-86.
See #Current Research section for a list of journals for additional information on the topic of flooded soils.
Edited by students of Kate Scow






