Silver as an Antimicrobial Agent: Difference between revisions
No edit summary |
No edit summary |
||
| Line 6: | Line 6: | ||
==Mechanism of action== | ==Mechanism of action== | ||
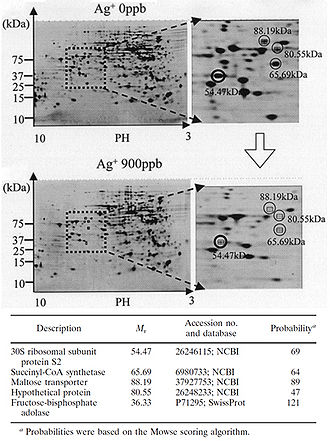
[[Image:FIGURE 2 Yamanaka et al..jpeg|thumb|330px|right|Figure 2. Protein expression is downregulated when bacteria are exposed to Ag+. 2-D polyacrylamide gel electrophoresis protein patterns obtained from the suspensions of E. coli after reactions with 0-ppb and 900-ppb Ag+ solutions for 3 h. Some spots indicate decreased amounts of protein expression after reaction with a 900-ppb solution, compared to results after reaction with a 0-ppb solution. Four spots with a decrease to less than one-third of the original amount are marked with open circles. The circled spots were cut out from the gel and analyzed with MALDI-TOF mass spectrometry. The probable identity of the proteins represented by the circled spots were determined by the NCBI and Swiss-Prot databases. Fructose-bisphosphate adolase was identified as another possible downregulated protein through this same process (not shown on this gel). The identity of the protein marked "Hypothetical protein" could not be determined through protein databases (Yamanaka et al. 2005).]][[Image:FIGURE 3 Feng et al.jpg|thumb|500px|right|Figure 3. Treatment of cells with Ag+ results in DNA condensation, cell wall damage, and silver granule formation. (A) E. coli and (B) S. aureus cells with and without Ag+ treatment were observed with TEM (Feng et al. 2000).]][[Image:FIGURE 4 Guggenbichler et al.jpg|thumb|400px|right|Figure 4. Treatment with silver leads to dehydration of microbial cells. A) Staphylococcus aureus without silver treatment as found on a catheter. B) Staphylococcus aureus on a silver-containing material with microdispersed silver particles throughout the matrix. Notice the shrunken appearance of the cells (Guggenbichler et al. 1999).]][[Image:FIGURE 5 Structure of silver sulfadiazine.jpg|thumb|250px|right|Figure 5. Structure of silver sulfadiazine.]][[Image:FIGURE 6 Fox and Modak.jpg|thumb|300px|right|Figure 6. | [[Image:FIGURE 2 Yamanaka et al..jpeg|thumb|330px|right|Figure 2. Protein expression is downregulated when bacteria are exposed to Ag+. 2-D polyacrylamide gel electrophoresis protein patterns obtained from the suspensions of E. coli after reactions with 0-ppb and 900-ppb Ag+ solutions for 3 h. Some spots indicate decreased amounts of protein expression after reaction with a 900-ppb solution, compared to results after reaction with a 0-ppb solution. Four spots with a decrease to less than one-third of the original amount are marked with open circles. The circled spots were cut out from the gel and analyzed with MALDI-TOF mass spectrometry. The probable identity of the proteins represented by the circled spots were determined by the NCBI and Swiss-Prot databases. Fructose-bisphosphate adolase was identified as another possible downregulated protein through this same process (not shown on this gel). The identity of the protein marked "Hypothetical protein" could not be determined through protein databases (Yamanaka et al. 2005).]][[Image:FIGURE 3 Feng et al.jpg|thumb|500px|right|Figure 3. Treatment of cells with Ag+ results in DNA condensation, cell wall damage, and silver granule formation. (A) E. coli and (B) S. aureus cells with and without Ag+ treatment were observed with TEM (Feng et al. 2000).]][[Image:FIGURE 4 Guggenbichler et al.jpg|thumb|400px|right|Figure 4. Treatment with silver leads to dehydration of microbial cells. A) Staphylococcus aureus without silver treatment as found on a catheter. B) Staphylococcus aureus on a silver-containing material with microdispersed silver particles throughout the matrix. Notice the shrunken appearance of the cells (Guggenbichler et al. 1999).]][[Image:FIGURE 5 Structure of silver sulfadiazine.jpg|thumb|250px|right|Figure 5. Structure of silver sulfadiazine.]][[Image:FIGURE 6 Fox and Modak.jpg|thumb|300px|right|Figure 6. Percent completion of reactions of various silver compounds with human serum. Human serum was incubated with 10 µmol of each silver salt. At the intervals designated, portions were removed and centrifuged, and the clear supernatants were analyzed to determine the amount of unreacted silver compound. The amount of compound used was taken on 100%. (Fox and Modak 1974).]]<br>Although the antimicrobial properties of silver have been known for centuries, we have only recently begun to understand the mechanisms by which silver inhibits bacterial growth. It is thought that silver atoms bind to thiol groups (-SH) in enzymes and subsequently cause the deactivation of enzymes. Silver forms stable S-Ag bonds with thiol-containing compounds in the cell wall that are involved in transmembrane energy generation and ion transport (Klueh et al. 2000). It is also believed that silver can take part in catalytic oxidation reactions that result in the formation of disulfide bonds (-R-S-S-R-). Silver does this by catalyzing the reaction between oxygen molecules in the cell and hydrogen atoms of thiol groups: water is released as a product and two thiol groups become covalently bonded to one another through a disulfide bond (Davies and Etris 1997). The silver-catalyzed formation of disulfide bonds could possibly change the shape of cellular enzymes and subsequently affect their function. <br> | ||
<br>The silver-catalyzed formation of disulfide bonds can lead to changes in protein structure and the inactivation of key enzymes, such as those needed for cellular respiration (Davies and Etris 1997). 30S ribosomal subunit protein, succinyl coenzyme A synthetase, maltose transporter (MalK), and fructose bisphosphate adolase were identified with high probability as proteins whose expression decreases once cells are treated with a 900-ppb Ag+ solution (Yamanaka et al. 2005; Figure 2). It is hypothesized that silver ions bind to the 30S ribosomal subunit, deactivating the ribosome complex and preventing translation of proteins (Yamanaka et al. 2005). The proteins that were found to be downregulated upon treatment with Ag+ serve important functions to the cell: succinyl coenzyme A synthetase, an enzyme involved in the TCA cycle, catalyzes the conversion of succinyl-CoA to succinate while phosphorylating GDP to produce GTP; fructose bisphosphate adolase is an enzyme involved in glycolysis that catalyzes the breakdown of fructose-1,6-bisphosphate into glyceraldehyde-3-phosphate and dihydroxyacetone phosphate; MalK is a cytoplasmic membrane-associated protein involved in the transport of maltose (Bavoil et al. 1980). In one way or another, all of these proteins play a role in energy and ATP production for the cell, so the decreased expression of any one of these proteins could lead to cell death (Yamanaka et al. 2005).<br> | <br>The silver-catalyzed formation of disulfide bonds can lead to changes in protein structure and the inactivation of key enzymes, such as those needed for cellular respiration (Davies and Etris 1997). 30S ribosomal subunit protein, succinyl coenzyme A synthetase, maltose transporter (MalK), and fructose bisphosphate adolase were identified with high probability as proteins whose expression decreases once cells are treated with a 900-ppb Ag+ solution (Yamanaka et al. 2005; Figure 2). It is hypothesized that silver ions bind to the 30S ribosomal subunit, deactivating the ribosome complex and preventing translation of proteins (Yamanaka et al. 2005). The proteins that were found to be downregulated upon treatment with Ag+ serve important functions to the cell: succinyl coenzyme A synthetase, an enzyme involved in the TCA cycle, catalyzes the conversion of succinyl-CoA to succinate while phosphorylating GDP to produce GTP; fructose bisphosphate adolase is an enzyme involved in glycolysis that catalyzes the breakdown of fructose-1,6-bisphosphate into glyceraldehyde-3-phosphate and dihydroxyacetone phosphate; MalK is a cytoplasmic membrane-associated protein involved in the transport of maltose (Bavoil et al. 1980). In one way or another, all of these proteins play a role in energy and ATP production for the cell, so the decreased expression of any one of these proteins could lead to cell death (Yamanaka et al. 2005).<br> | ||
<br>Another one of the suggested mechanisms of the antimicrobial activity of silver was proposed by Klueh et al. (2000). It was proposed that Ag+ enters the cell and intercalates between the purine and pyrimidine base pairs disrupting the hydrogen bonding between the two anti-parallel strands and denaturing the DNA molecule (Klueh et al. 2000). Although this has yet to be proved, it has been shown that silver ions do associate with DNA once they enter the cell (Fox and Modak 1974). <br> | <br>Another one of the suggested mechanisms of the antimicrobial activity of silver was proposed by Klueh et al. (2000). It was proposed that Ag+ enters the cell and intercalates between the purine and pyrimidine base pairs disrupting the hydrogen bonding between the two anti-parallel strands and denaturing the DNA molecule (Klueh et al. 2000). Although this has yet to be proved, it has been shown that silver ions do associate with DNA once they enter the cell (Fox and Modak 1974). <br> | ||
Revision as of 22:10, 14 April 2010
By: Gian Garduque
Introduction
Mechanism of action
Although the antimicrobial properties of silver have been known for centuries, we have only recently begun to understand the mechanisms by which silver inhibits bacterial growth. It is thought that silver atoms bind to thiol groups (-SH) in enzymes and subsequently cause the deactivation of enzymes. Silver forms stable S-Ag bonds with thiol-containing compounds in the cell wall that are involved in transmembrane energy generation and ion transport (Klueh et al. 2000). It is also believed that silver can take part in catalytic oxidation reactions that result in the formation of disulfide bonds (-R-S-S-R-). Silver does this by catalyzing the reaction between oxygen molecules in the cell and hydrogen atoms of thiol groups: water is released as a product and two thiol groups become covalently bonded to one another through a disulfide bond (Davies and Etris 1997). The silver-catalyzed formation of disulfide bonds could possibly change the shape of cellular enzymes and subsequently affect their function.
The silver-catalyzed formation of disulfide bonds can lead to changes in protein structure and the inactivation of key enzymes, such as those needed for cellular respiration (Davies and Etris 1997). 30S ribosomal subunit protein, succinyl coenzyme A synthetase, maltose transporter (MalK), and fructose bisphosphate adolase were identified with high probability as proteins whose expression decreases once cells are treated with a 900-ppb Ag+ solution (Yamanaka et al. 2005; Figure 2). It is hypothesized that silver ions bind to the 30S ribosomal subunit, deactivating the ribosome complex and preventing translation of proteins (Yamanaka et al. 2005). The proteins that were found to be downregulated upon treatment with Ag+ serve important functions to the cell: succinyl coenzyme A synthetase, an enzyme involved in the TCA cycle, catalyzes the conversion of succinyl-CoA to succinate while phosphorylating GDP to produce GTP; fructose bisphosphate adolase is an enzyme involved in glycolysis that catalyzes the breakdown of fructose-1,6-bisphosphate into glyceraldehyde-3-phosphate and dihydroxyacetone phosphate; MalK is a cytoplasmic membrane-associated protein involved in the transport of maltose (Bavoil et al. 1980). In one way or another, all of these proteins play a role in energy and ATP production for the cell, so the decreased expression of any one of these proteins could lead to cell death (Yamanaka et al. 2005).
Another one of the suggested mechanisms of the antimicrobial activity of silver was proposed by Klueh et al. (2000). It was proposed that Ag+ enters the cell and intercalates between the purine and pyrimidine base pairs disrupting the hydrogen bonding between the two anti-parallel strands and denaturing the DNA molecule (Klueh et al. 2000). Although this has yet to be proved, it has been shown that silver ions do associate with DNA once they enter the cell (Fox and Modak 1974).
Most of the proposed mechanisms involve silver entering the cell in order to cause damage. How would a metal like silver, or its ionized form Ag+, get across the hydrophobic cellular membrane to access the cytoplasm? From the perspective of a transmembrane protein, the silver ion simply appears to be a particle of certain size with a +1 charge. It is possible that silver ions get access to the interior of cells through transmembrane proteins that normally function to transport ions other than silver ions. Transmembrane proteins such as CopB-ATPase from Enterococcus hirae have been shown to be able to transport silver ions although its putative function is a copper transporter (Solioz and Odermatt 1995). This proves that there are ways for silver to be transported across the cell membrane even though specific silver transporters may not exist.
In order for silver to have any antimicrobial properties, it must be in its ionized form (Lok et al, 2007; Rai et al. 2009). Silver in its unionized form is inert (Guggenbichler et al. 1999), but contact with moisture leads to the release of silver ions (Radheshkumar and Munstedt 2005). Thus, all forms of silver or silver containing compounds with observed antimicrobial properties are in one way or another sources of silver ions (Ag+); these silver ions may be incorporated into the substance and released slowly with time as with silver sulfadiazine, or the silver ions can come from ionizing the surface of a solid piece of silver as with silver nanoparticles.
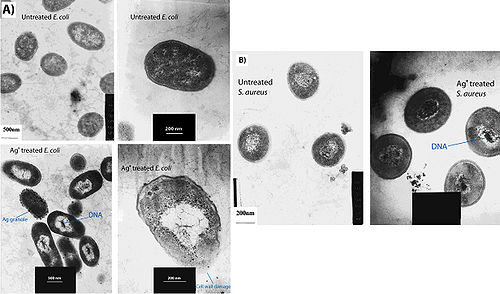
Feng et al. (2000) conducted a study to observe the effects of silver ions on gram-positive and gram-negative bacteria, namely Staphylococcus aureus and Escherichia coli. They treated cells with AgNO3, which is source of Ag+ in aqueous environments, and looked at the structural and morphological effects of these silver ions on the cells. The cells were exposed to AgNO3 for 4-12 hours before being prepared for microscopy. The cell were then fixed and sliced with an ultramicrotome to produce ultrathin sections for transmission electron microscopy (TEM). They observed that cells exposed to the Ag+ ions seemed to have activated a stress response that led to the condensation of DNA in the center of the cell. They also observed cell membrane detachment from the cell wall, cell wall damage, and electron dense granules outside and, in some instances, inside the cell (Figure 3). It was proposed that condensation of DNA occurred as a protective measure in order to protect the genetic information of the cell (Feng et al. 2000), however condensation of DNA could also prevent cell replication by preventing the DNA from being accessed by transcriptional enzymes such as DNA polymerase. The electron dense granules that formed inside and outside the cell were extracted and subjected to X-ray microanalysis to determine their composition. It was discovered that the granules were in part composed of silver and sulfur. This finding supports the idea that silver inactivates proteins by binding to sulfur-containing compounds (Klueh et al. 2000). It was also observed that when treated with Ag+, E. coli, a gram-negative bacterium, sustained more structural damages than the gram-positive S. aureus (Feng et al. 2000).
There are two explanations as to why gram-positive bacteria are less susceptible to Ag+ than gram-negative bacteria. The first involves the charge of peptidoglycan molecules in the bacterial cell wall. Gram-positive bacteria have more peptidoglycan than gram-negative bacteria because of their thicker cell walls, and because peptidoglycan is negatively charged and silver ions are positively charged, more silver may get trapped by peptidoglycan in gram-positive bacteria than in gram-negative bacteria (Kawahara et al. 2000). The decreased susceptibility of gram-positive bacteria can also simply be explained by the fact that the cell wall of gram-positive bacteria is thicker than that of gram-negative bacteria.
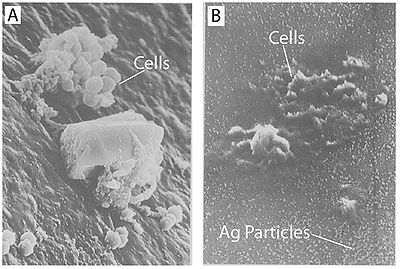
It has also been shown that treating cells with silver leads to cell shrinkage and dehydration (Figure 4) (Guggenbichler et al. 1999). The TEM images from Feng et al. (2000) (Figure 4) show that cells that sustained extensive damage eventually ended up with cell wall and cell membrane damage. Damage to the cell membrane could lead to the leaking of cytoplasm from the cell, which would result in dehydrated and shrunken cells as shown by the SEM images from Guggenbichler et al. (1999).
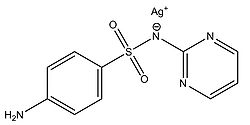
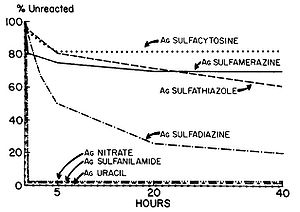
Fox and Modak (1974) explored the mechanism of prevention of burn wound infections by silver sulfadiazine. At the time of publication, it had been known for quite a while that silver sulfadiazine (Figure 5) delivered in the form of a topical cream was effective at preventing infections in burn wounds, however it was not known if the antimicrobial activity was due entirely to the silver ions or if the sulfadiazine anion also contributed to the bactericidal effect. Tests from the study showed that sulfadiazine from silver sulfadiazine does not get transported into cells as much as silver. Silver isotopes (110Ag+) were also used to show that silver ions that enter cells complex with DNA. Additionally, the rate at which certain silver containing compounds release silver ions into solution was measured by adding silver compounds to human serum and measuring the amount of unreacted silver compound with increasing time (Figure 6). Notice in Figure 6 how silver sulfadiazine (Ag sulfadiazine) gradually releases the majority of its silver ions into solution over an extended period of time whereas silver nitrate immediately released all of its silver ions into solution (Fox and Modak 1974). Therefore, silver sulfadiazine's effectiveness as an antimicrobial agent for preventing burn wound infections is due to its tendency to dissociate in solution: silver sulfadiazine provides a steady supply of silver ions over a long period of time where as other silver salts such as silver nitrate release a large amount of silver ions all at once. If silver is employed as the primary antimicrobial agent in burn wound creams, the burn wound needs a steady supply of silver ions over a long period of time to kill off any microbes that could possibly infect the wound until it heals. Compounds that release silver ions all at once would need to be applied very frequently in order to kill off invading bacteria and prevent infection, and sometimes highly frequent application isn't always practical or possible for individuals, so compounds that constantly release a bactericidal amount of silver ions, such as silver sulfadiazine, are the most effective at preventing burn wound infections.
Section 2
Include some current research, with at least one figure showing data.
Section 3
Include some current research, with at least one figure showing data.
Conclusion
Overall text length at least 3,000 words, with at least 3 figures.
References
Edited by Gian Garduque,student of Joan Slonczewski for BIOL 238 Microbiology, 2010, Kenyon College.