User:Using Yeast to Convert Carbon Dioxide Gas to Carbonates For Building Materials: Difference between revisions
| Line 13: | Line 13: | ||
<br> <br> | <br> <br> | ||
== | ==Carbon Sequestration== | ||
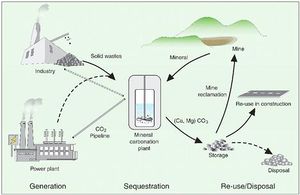
<br> | <br>There have been many efforts to somehow channel this increasing amount of atmospheric CO2 to more harmless or even more helpful forms. One method is carbon sequestration, in which CO2 is captured from industrial facilities, transported via pipes and then injected underground deep into porous rocks underground which are then blocked from the top by non-porous rock (EPA, 2013). This essentially traps the CO2 underground permanently. This sequestration can pose some geological safety and security concerns and there are a limited amount of sites that can safely store CO2 and these sites must then be monitored to ensure there is no leakage (EPA, 2013). | ||
Some MIT researchers have recently developed a new method to capture carbon dioxide while also putting it to good use. They have used baker’s yeast, or Saccharomyces cerevisiae, to convert carbon dioxide gas into carbonates useful as building materials (Trafton, 2010). After capturing the carbon dioxide in water, it is then combined with mineral ions to form solid carbonates (Trafton, 2010). Although yeast do not perform this conversion on their own, they have been genetically engineered by these researchers to express genes found in snails such as the albalone who build their own hard shells composed of calcium carbonate from carbon dioxide and mineral ions found in their marine habitats (Trafton, 2010). The genes they have utilized code for enzymes that facilitate the mineralization of carbon dioxide (Trafton, 2010). This experiment holds promise as it “can produce about two pounds of carbonate for every pound of carbon dioxide captured” and “captures carbon dioxide at a higher rate” than processes companies have used to convert CO2 to solid building material (Trafton, 2010). | |||
Bricks made from CO2 would potentially solve two problems- sequester excess atmospheric CO2 and eliminate additional CO2 emissions that result from traditional brick-making. Although this team of researchers are not the first to propose CO2 sequestration, they do seem to have developed an efficient technique. “It involves no chemicals or temperature changes, unlike chemical solidification methods” (Trafton, 2010). The researchers want to make the process efficient enough to deal with large amounts of CO2 to be useful in an industrial setting such as a factory or power plant. | |||
<br> | |||
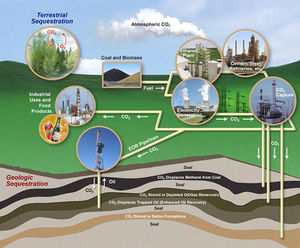
[[Image:Sequestration1.jpeg|thumb|300px|right|Outlining the Carbon Storage Program in the United States. http://fossil.energy.gov/programs/sequestration/overview.html]] | [[Image:Sequestration1.jpeg|thumb|300px|right|Outlining the Carbon Storage Program in the United States. http://fossil.energy.gov/programs/sequestration/overview.html]] | ||
==Section 2== | ==Section 2== | ||
Revision as of 03:34, 25 April 2013
Carbon Cycle
There are many carbon reservoirs in the environment, but one of particular concern is CO2 which is found in the atmosphere. Although the amount of carbon found in the atmosphere in the form of CO2 is much less than that found in the reservoirs found in the ocean or in land, the burning of fossil fuels has increased the balance of atmospheric CO2. Also, although the ocean acts as a buffer by absorbing some excess CO2, atmospheric CO2 “continues to increase at a rate of about 1% per year” (Slonczewski & Foster, p 833). This is problematic because CO2 is a greenhouse gas and this means it can absorb solar radiation and thereby heat up the atmosphere. Therefore, CO2 is contributing to gradual global warming. (Slonczewski & Foster, p 833) Humans have altered the balance of atmospheric CO2 in the environment. Industrial activities have increases the amount of CO2 release globally due largely to the burning of fossil fuels, which are hydrocarbons formed from the organic remains of prehistoric plants and animals that when burned release CO2 as a byproduct (Slonczewski & Foster, p 834). Additionally, deforestation contributes largely to increasing atmospheric CO2 content due to burning wood and especially due to microbial decomposition of chopped down trees (Slonczewski & Foster, p834).
Carbon Sequestration
There have been many efforts to somehow channel this increasing amount of atmospheric CO2 to more harmless or even more helpful forms. One method is carbon sequestration, in which CO2 is captured from industrial facilities, transported via pipes and then injected underground deep into porous rocks underground which are then blocked from the top by non-porous rock (EPA, 2013). This essentially traps the CO2 underground permanently. This sequestration can pose some geological safety and security concerns and there are a limited amount of sites that can safely store CO2 and these sites must then be monitored to ensure there is no leakage (EPA, 2013).
Some MIT researchers have recently developed a new method to capture carbon dioxide while also putting it to good use. They have used baker’s yeast, or Saccharomyces cerevisiae, to convert carbon dioxide gas into carbonates useful as building materials (Trafton, 2010). After capturing the carbon dioxide in water, it is then combined with mineral ions to form solid carbonates (Trafton, 2010). Although yeast do not perform this conversion on their own, they have been genetically engineered by these researchers to express genes found in snails such as the albalone who build their own hard shells composed of calcium carbonate from carbon dioxide and mineral ions found in their marine habitats (Trafton, 2010). The genes they have utilized code for enzymes that facilitate the mineralization of carbon dioxide (Trafton, 2010). This experiment holds promise as it “can produce about two pounds of carbonate for every pound of carbon dioxide captured” and “captures carbon dioxide at a higher rate” than processes companies have used to convert CO2 to solid building material (Trafton, 2010).
Bricks made from CO2 would potentially solve two problems- sequester excess atmospheric CO2 and eliminate additional CO2 emissions that result from traditional brick-making. Although this team of researchers are not the first to propose CO2 sequestration, they do seem to have developed an efficient technique. “It involves no chemicals or temperature changes, unlike chemical solidification methods” (Trafton, 2010). The researchers want to make the process efficient enough to deal with large amounts of CO2 to be useful in an industrial setting such as a factory or power plant.
Section 2
Include some current research in each topic, with at least one figure showing data.
Section 3
Include some current research in each topic, with at least one figure showing data.
Conclusion
Overall paper length should be 3,000 words, with at least 3 figures.
References
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2009, Kenyon College.