Flavivirus NS1 protein: Difference between revisions
| Line 8: | Line 8: | ||
==Basic Flavivirus Structure and Maturation== | ==Basic Flavivirus Structure and Maturation== | ||
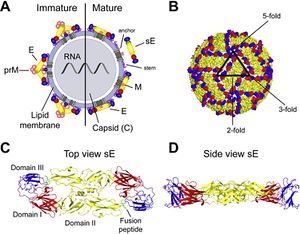
[[Image:Alex Fig 1.jpeg|thumb|300px|right|Figure 1. Flavivirus Structure. (A) Schematic representation of a flavivirus particle. Left: immature virion; right: mature virion | [[Image:Alex Fig 1.jpeg|thumb|300px|right|Figure 1. Flavivirus Structure. (A) Schematic representation of a flavivirus particle. Left: immature virion; right: mature virion.(B) Herringbone-like arrangement of 90 E protein dimers at the virion surface as determined by cryo-electron microscopy.]] | ||
<br> Flaviviruses are small and have only seven nonstructural and three structural proteins. These structural proteins are designated E, prM/M, and C for envelope, precursor of the membrane or membrane, and capsid respectively (Fig 1). Immature virions are formed by budding of the endoplasmic reticulum. These non-infections virions form prM-E heterodimers and in order to mature into infectious virions, these heterodimers need to be cleaved. This allows the E proteins to form herringbone like antiparallel dimers and therefore rearranges the whole viral envelope into a smoother surface (Heinz and Stiasny 2012). | <br> Flaviviruses are small and have only seven nonstructural and three structural proteins. These structural proteins are designated E, prM/M, and C for envelope, precursor of the membrane or membrane, and capsid respectively (Fig 1). Immature virions are formed by budding of the endoplasmic reticulum. These non-infections virions form prM-E heterodimers and in order to mature into infectious virions, these heterodimers need to be cleaved. This allows the E proteins to form herringbone like antiparallel dimers and therefore rearranges the whole viral envelope into a smoother surface (Heinz and Stiasny 2012). | ||
Revision as of 19:07, 25 April 2014
Flavivirus
By Alex Gonzales
Flaviviruses are small, positive sense RNA viruses that can cause yellow fever, dengue, Japanese encephalitis, and West Nile virus in addition to tick-borne encephalitis (Hetnz and Stiasny 2012). These pathogens use arthropods such as mosquitoes from the Aedes genus as vectors and are found in temperate and tropical areas. The specificity of their natural hosts means that flaviviruses all have a certain endemic/ epidemic area rather than being equally distributed around the globe. However, that also means that the viruses range can expand when the host adapts to new places. For example the introduction of WN vector and the subsequent outbreak in New York in 1999 lead to the expansion of its target range into North and South America (Vasilakis et al. 2011).
Globally, the dengue virus has the most impact with about 50-100 million infections per year which result in more than 20,000 deaths annually. This is probably due to the fact that there are vaccines for yellow fever, Japanese encephalitis, and tick born encephalitis but an effective dengue vaccine has yet to be discovered. The delay in the dengue vaccine comes from concerns that the vaccines might actually predispose the subject to infections from other strains of dengue virus (Heintz, Stiasny 2012).
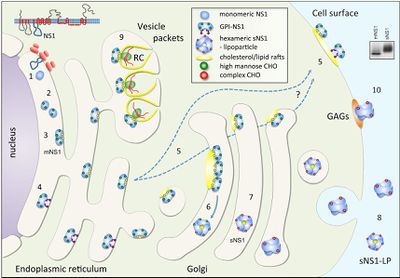
In the search for a viable vaccine, over three decades of research has been focused on the structure and pathology of Flaviviruses. Over the course of their work, scientists have discovered much, but many questions still remain about an enigmatic nonstructural glycoprotein, NS1. NS1 was discovered in 1970 in the serum of a patients infected with dengue and it was later discovered that all flaviviruses NS1 genes were highly homologous. Additionally, it was found that NS1 has multiple oligomeric forms a dimer and a hexamer, which are involved in replication and host immune system evasion respectively. It has also been suggested that the two main forms of NS1 are involved in both protection and pathogenesis of their host (Muller, Young 2013).
Basic Flavivirus Structure and Maturation
Flaviviruses are small and have only seven nonstructural and three structural proteins. These structural proteins are designated E, prM/M, and C for envelope, precursor of the membrane or membrane, and capsid respectively (Fig 1). Immature virions are formed by budding of the endoplasmic reticulum. These non-infections virions form prM-E heterodimers and in order to mature into infectious virions, these heterodimers need to be cleaved. This allows the E proteins to form herringbone like antiparallel dimers and therefore rearranges the whole viral envelope into a smoother surface (Heinz and Stiasny 2012).
E proteins are important for cell infection as they mediate binding to cell receptors and fusion with endosomal membranes after host cell endocytosis. To help fusion with the endosomal membrane, E protiens undergo yet another structural change triggered by pH. In low pH, the E protein dimer disassociates, and becomes a trimer with an exposed fusion peptide at one tip. Due to the essential role E proteins play in entering the host cell, they are a main target of anti-flivivrus antibodies which attempt to block entry into the cell by hindering attachment and endocytosis or membrane fusion (Heinz and Stiasny 2012).
NS1 Structure