Desulfatibacillum alkenivorans: Difference between revisions
No edit summary |
No edit summary |
||
| Line 7: | Line 7: | ||
Ground water is also contaminated by pollutants that are not highly toxic, but can be utilized or modified by microorganisms to become more toxic. For instance over-fertilization in agriculture leads to an increased nitrate concentration which i.e. can cause the Blue Baby syndrome. This is seen in infants younger than six month old who rely on bacteria to digest their food. Some of these bacteria also convert nitrate, a component of fertilizer, to nitrite. In the blood nitrite reacts with hemoglobin interfering with its ability to carry oxygen. The babies show sign of suffocation and gets a bluish skin [[#References|[2]]]. | Ground water is also contaminated by pollutants that are not highly toxic, but can be utilized or modified by microorganisms to become more toxic. For instance over-fertilization in agriculture leads to an increased nitrate concentration which i.e. can cause the Blue Baby syndrome. This is seen in infants younger than six month old who rely on bacteria to digest their food. Some of these bacteria also convert nitrate, a component of fertilizer, to nitrite. In the blood nitrite reacts with hemoglobin interfering with its ability to carry oxygen. The babies show sign of suffocation and gets a bluish skin [[#References|[2]]]. | ||
= | =Metabolism= | ||
==Autotrophy== | |||
<i>Desulfatibacillum alkenivorans</i> AK-01 can grow chemolithoautotrophically with H<sub>2</sub>, CO<sub>2</sub>, and sulfate [[#References|[10]]]. The [http://en.wikipedia.org/wiki/Wood%E2%80%93Ljungdahl_pathway Wood-Ljungdahl pathway] is the predicted mechanism of CO<sub>2</sub> fixation due the presence of all the required genes for the pathway and the absence of the gene for [http://en.wikipedia.org/wiki/ATP_citrate_lyase ATP citrate lyase], an important enzyme within the [http://en.wikipedia.org/wiki/Reverse_Krebs_cycle reverse TCA cycle] [[#References|[3]]]. The Wood-Ljungdahl pathway is used to synthesize cellular carbon from CO<sub>2</sub> while obtaining energy from the coupling of hydrogen oxidation to sulfate reduction [[#References|[3]]]. | |||
==Syntrophy== | ==Syntrophy== | ||
Revision as of 21:19, 12 December 2012
Desulfatibacillum alkenivorans AK-01
Description and Significance
Groundwater serves as water for more than 50% of people living in North America therefore a significant public resource. To date, major contamination of groundwater in North America are due to the release and use of chlorinated ethenes by industry. Examples of such toxic compounds are perchloroethene (PCE), trichloroethene (TCE). Carbon tetrachloride (CT) is also a major groundwater pollutant [4]. These compounds were widely used as solvents for dry cleaning and in textile manufacturing. They are sufficiently water soluble and can travel through soil where they reach the groundwater. The relative high concentration of them here can be harmful [6]. Ground water is also contaminated by pollutants that are not highly toxic, but can be utilized or modified by microorganisms to become more toxic. For instance over-fertilization in agriculture leads to an increased nitrate concentration which i.e. can cause the Blue Baby syndrome. This is seen in infants younger than six month old who rely on bacteria to digest their food. Some of these bacteria also convert nitrate, a component of fertilizer, to nitrite. In the blood nitrite reacts with hemoglobin interfering with its ability to carry oxygen. The babies show sign of suffocation and gets a bluish skin [2].
Metabolism
Autotrophy
Desulfatibacillum alkenivorans AK-01 can grow chemolithoautotrophically with H2, CO2, and sulfate [10]. The Wood-Ljungdahl pathway is the predicted mechanism of CO2 fixation due the presence of all the required genes for the pathway and the absence of the gene for ATP citrate lyase, an important enzyme within the reverse TCA cycle [3]. The Wood-Ljungdahl pathway is used to synthesize cellular carbon from CO2 while obtaining energy from the coupling of hydrogen oxidation to sulfate reduction [3].
Syntrophy
Desulfatibacillum alkenivorans AK-01 is capable of syntrophically metabolizing alkanes when coupled with Methanospirillum hungatei JF-1. M. hungatei is an anaerobic methanogen that utilizes formate or H2-CO2 [7]. When AK-01 was grown with M. hungatei JF-1 in a medium free of sulfate, methane was produced from n-hexadecane [3]. Cultures of M. hungatei JF-1 and D. alkenivorans AK-01 grown separately failed to produce methane [3]. The mechanism of syntrophic metabolism was predicted after the detection of three formate dehydrogenase complexes in the AK-01 genome. The formate dehydrogenase complexes are capable of producing formate from CO2 and H2, using electrons from a menaquinone pool produced during alkane metabolism. This suggests that formate production may be utilized to transfer electrons derived from alkanes to the methanogen [3].
Heterotrophy
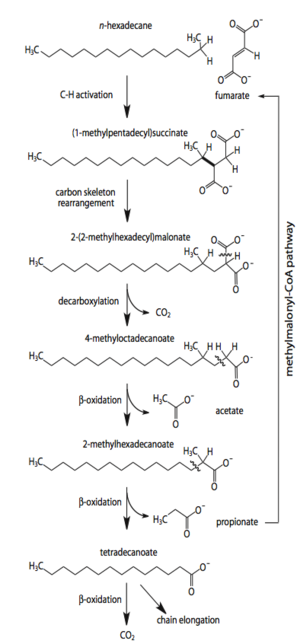
Desulfatibacillum alkenivorans AK-01 is capable of using sulfate, sulfite and thiosulfate, but not sulfur, as terminal electron acceptors during heterotrophic metabolism [10]. Alkane degradation is initiated through the subterminal addition of the alkane carbon to the double bond of fumarate [2]. The alkylsuccinate intermediate is further degraded through processes of carbon-skeleton rearrangement, decarboxylation, and beta-oxidation [1]. The resultant acetate molecule is further oxidized to CO2 through the reversal of the Wood-Ljungdahl pathway [3]. Strain AK-01 is able to completely oxidize alkanes to carbon dioxide when coupled with sulfate reduction [11]. This was shown through the formation of radiolabeled CO2 from radiolabeled [1-14C]hexadecane and the corresponding loss of sulfate that was congruent with the theoretical stoichiometric ratio [11]. Under sulfate-limiting conditions, only 3% of the total alkane present was degraded, demonstrating the dependence of alkane degradation on sulfate reduction [10]. When sulfate concentrations were sufficient, 91% of the alkane present was degraded [10]. The mechanism of initiation via addition to fumarate is not unique to D. alkenivorans AK-01. It has also been observed in the sulfate-reducer Desulfoglaeba alkanexedens [6], Desulfatibacillum aliphaticivorans CV2803 [5], and the denitrifying strain HxN1, a member of the Betaproteobacteria [14]. This reaction involving fumarate represents an alternative mechanism of alkane activation compared to the more commonly known hydroxylation reaction by monooxygenases in aerobic environments [11].
Ecology
From an evolutionary perspective, D. alkenivorans AK-01 is well adapted to occupy a variety of niches. The discovery of genes related to motility and oxygen detoxification suggests AK-01 may be able to adapt to a greater variety of environmental conditions than previously thought [3]. Furthermore, AK-01 is capable of exhibiting three distinct modes of metabolism, giving it a distinct advantage over more specialized organisms [3]. The microbial interactions of AK-01 in situ have yet to be analyzed. There are no known diseases associated with D. alkenivorans AK-01 [13].
Future Applications
Organisms capable of coupling alkane degradation with sulfate reduction play a key role in sites contaminated with hydrocarbons, as these sites typically become anoxic over time due to aerobic respiration and decomposition [4]. Additionally, sulfate is relatively abundant in marine waters compared to nitrate and iron, which typically exist at low concentrations [9]. In one study, analysis of sediment polluted with petroleum after 502 days showed strains most closely related to AK-01 were the most abundant [9], further emphasizing possible bio-remedial applications of D. alkenivorans AK-01 in the future. It has been experimentally shown that AK-01 can interact with methanogens to degrade alkanes syntrophically. This mechanism could be applied within the oil industry, as the current techniques used to retrieve fossil fuels are only able to extract 40% of the available resource [8]. The residual oil could be used to harvest methane gas through syntrophic degradation as an alternative energy source [8]. Current experiments expect a yield of 3 mmol methane gas per gram of residual oil. Taking into account the number of U.S. oil reservoirs, syntrophic alkane degradation could provide 17% of the natural gas consumed in the United States [8].
References
1. Callaghan, A., Gieg, L., Kropp, K., Suflita, J. and Young, L. “Comparison of mechanisms of alkane metabolism under sulfate-reducing conditions among two bacterial isolates and a bacterial consortium.” Appl Environ Microbiol, 2006, DOI: 10.1128/AEM02896-05
2. Callaghan, A., Wawrik, B., Ní Chadhain, S., Young, L. and Zylstra, G. “Anaerobic alkane-degrading strain AK-01 contains two alkylsuccinate synthase genes.” Biochem Biophys Res Commun, 2008, DOI: 10.1016/j.bbrc.2007.11.094
3. Callaghan, A., Morris, B., Pereira, I., McInerney, M., Austin, R., Groves, J., Kukor, J., Suflita, J., Young, L., Zylstra, G. and Wawrik, B. “The genome sequence of Desulfatibacillum alkenivorans AK-01: a blueprint for anaerobic alkane oxidation.” Environ Microbiol, 2012, DOI: 10.1111/j.1462-2920.2011.02516.x
4. Canfield, D., Jorgensen, B., Fossing, H., Glud, R., Gundersen, J., Ramsing, N., Thamdrup, B., Hansen, J., Nielsen, L. and Hall, P. “Pathways of organic carbon oxidation in three continental margin sediments.” Mar Geol, 1993, DOI: 10.1016/0025-3227(93)90147-N
5. Cravo-Laureau, C., Matheron, R., Cayol, J., Joulian, C. and Hirschler-Réa, A. “Desulfatibacillum aliphaticivorans gen. nov., sp. nov., an n-alkane- and n-alkene-degrading, sulfate-reducing bacterium.” Int J Syst Evol Microbiol, 2004, DOI: 10.1099/ijs.0.02717-0
6. Davidova, I., Duncan, K., Choi, O. and Suflita, J. “Desulfoglaeba alkanexedens gen. nov., sp. nov., an n-alkane-degrading, sulfate-reducing bacterium.” Int J Syst Evol Microbiol, 2006, DOI: 10.1099/ijs.0.64398-0
7. Ferry, J., Smith, P. and Wolfe, R. “Methanospirillum, new genus of methanogenic bacteria, and characterization of Methanospirillum hungatei sp. nov.” Int J Syst Evol Microbiol, 1974, Retrieved from http://ijs.sgmjournals.org/content/24/4/465
8. Gieg, L., Duncan, K. and Suflita, J. “Bioenergy Production via Microbial Conversion of Residual Oil to Natural Gas.” Appl Environ Microbiol, 2008, DOI: 10.1128/AEM.00119-08
9. Miralles, G., Grossi, V., Acquaviva, M., Duran, R., Bertrand, J. and Cuny, P. “Alkane biodegradation and dynamics of phylogenetic subgroups of sulfate-reducing bacteria in an anoxic costal marine sediment artificially contaminated with oil.” Chemosphere, 2007, DOI: 10.1016/j.chemosphere.2007.01.033
10. So, C. and Young, L. “Isolation and characterization of a sulfate-reducing bacterium that anaerobically degrades alkanes.” Appl Environ Microbiol, 1999, Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC91444/
11. So, C. and Young, L. “Initial reactions in anaerobic alkane degradation by a sulfate reducer, strain AK-01.” Appl Environ Microbiol, 1999, Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC91754/
12. So, C., Phelps, C. and Young, L. “Anaerobic transformation of alkanes to fatty acids by a sulfate-reducing bacterium, strain Hxd3.” Appl Environ Microbiol, 2003, DOI: 10.1128/AEM.69.7.3892-3900.2003
13. U.S. Department of Energy Joint Genome Institute. “Desulfatibacillum alkenivorans AK-01.” 2010, Retrieved from http://genome.jgi.doe.gov/desa1/desa1.info.html
14. Wilkes, H., Rabus, R. and Fischer, T. “Anaerobic degradation of n-hexane in a denitrifying bacterium: Further degradation of the initial intermediate (1-methylpentyl)succinate via C-skeleton rearrangement.” Arch Microbiol, 2002, DOI: 10.1007/s00203-001-0381-3