Bacteriophages in Cancer Biology and Treatment
Section
By Salome Shubitidze!
At right is a sample image insertion. It works for any image uploaded anywhere to MicrobeWiki.
The insertion code consists of:
Double brackets: [[
Filename: F2.large.jpg
Thumbnail status: |thumb|
Pixel size: |300px|
Placement on page: |right|
Legend/credit: Electron micrograph of the Ebola Zaire virus. This was the first photo ever taken of the virus, on 10/13/1976. By Dr. F.A. Murphy, now at U.C. Davis, then at the CDC. Every image requires a link to the source.
Closed double brackets: ]]
Other examples:
Yes that does seem to be the case with such viruses
Italic
Subscript: H2O
Superscript: Fe3+
Introduce the topic of your paper. What is your research question? What experiments have addressed your question? Applications for medicine and/or environment?
Sample citations: [1]
[2]
A citation code consists of a hyperlinked reference within "ref" begin and end codes.
To repeat the citation for other statements, the reference needs to have a names: "<ref name=aa>"
The repeated citation works like this, with a back slash.[1]
The use of Bacteriophages for drug delivery
Chemotherapy drugs, while shown to have anti-tumor effects, tend to result in severe toxicity and widespread distribution throughout the body: notably damaging healthy and malignant cells. New research has started to focus on using bacteriophages as an individualized drug-carrying anti-cancer therapy. The therapy would be targeted, based on genetically-modifying and chemically manipulating filamentous bacteriophages. In Bar et al. 2008, the phages were modified to display a host-specificity-conferring ligand, and carry a cytotoxic drug by chemical conjugation
Antibodies anti ErbB2 and anti ERGR were used to direct bacteriophages to cancer cells. ErbB2 is a a well known protein that is found to be over expressed in approximately 20% of invasive breast cancers. ERGR is a receptor protein that is found on the surface of cells that causes some cells to duplicate if an epidermal growth factor binds to it. They both belong to the epidermal growth factor receptor (EGFR) family (19). They are both common proteins that tend to be associated with the growth of cancers. The cell lines used in Bar et al. 2008 were SKBR3 and MDA-MB23 (human breast carcinoma cell lines), A431 (human epidermoid carcinoma), and HEK293 (human kidney). Phages were engineered to be linked to respective antibodies.
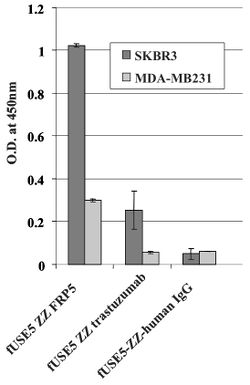
Assessing The Ability of the Engineered Antibody-Phages to go to Their Respective Receptors
A whole cell ELISA was performed in order to assess the binding of phage-attached antibodies (see figure 1.) MDA-MB231 cells express a low level of ErbB2 proteins, while SKBR3 express a much higher level of ErbB2 proteins. As shown, one can see that the antibody-complexed phages showed cell-specific binding. The engineered fUSE5-ZZ-chFRP5 phage had higher rates of binding to receptors on SKBR3 and even MDA-MB231 than the control phage fUSE5-ZZ-human IgG. This shows that the engineered phages are capable of attaching to their respective receptors through the use of antibodies.
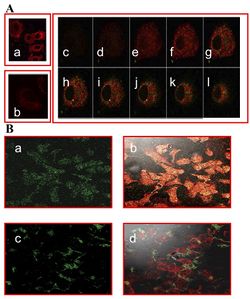
Assessing Engineered Antibody-Phages as Drug Carriers
In order to check if phages could effectively deliver drugs, researchers had to verify that they could enter into host cells. One way this was done was by visualizing phage internalization using confocal microscopy and immunofluorescence staining (See Figure 2). Bar et al. 2008 engineered its phages to carry hygromycin inside cancer cells. Hygromycin is an antibiotic produced by the bacterium Streptomyces hygroscopicus. It kills bacteria, fungi, and interestingly inhibits the growth of some eukaryotic cells. Therefore, by engineering phages to carry the this drug into cancer cells, it is possible further growth can be stopped. The assay was done at 2, 12, and 24 hours in order to track the rate at which phages entered malignant cells. Surprisingly, internalization of hygromycin was the greatest at 2 hours, and diminished over time. Figure 2, images "c" and "d" show the attempt of internalization of hygromycin conjugated phages complexed with with human IgG. They were unsuccessful, filling their role as controls. Therefore, data supports that engineered antibody carrying phages are capable of transporting large amounts of shipments of a drug into a compromised cell.
Changing the Tumor Microenvironment
Include some current research, with at least one figure showing data.
Phage Display Methods and Tumor-specific Antibody–receptor Pairs
Include some current research, with at least one figure showing data.
Activating the Innate Immune System
Tumors have become increasingly more clever in their ways of evading the immune system.
In both animal and human models, tumors have been discovered to actively suppress and manipulate immune responses. Usually our immune system's CD8+ cytotoxic T cells (CTL) and CD4 + helper T (Th)1 cells are capable of warding off cancer development by producing interferons (IFN)-�and cytotoxins (SCB). Cytotoxic T cells are a type of white blood cell that kill cancer cells, or cells that are dysfunctional. Interferons are signaling proteins released by infected host cells that alert nearby cells to strengthen up their defenses. The continual production of these type of cells leads to chronic inflammation, which has been tied to the spread of cancer. Therefore, evidence shows that when our immune system's initial responses fail to eradicate tumor cells, the flood of immunoregulatory cells leads to the resurgence of a "smarter" cancer.
Tumors have found a way to remain "hidden" from immune cells by promoting the secretion of immunomodulatory factors within the tumor microenvironment. CD4+ CD25+ FoxP3+ regulatory T cells (Tregs)are just a few examples of suppressive cells that aid a tumor's immune evasion. Regulatory T cells generally turn the immune response down.
There are many protein factors released by tumor cells that may recruit tumor-associated macrophages (TAMs). TAMs are M2- polarized. M2- polarized macrophages function to repair damaged tissues, and aid the final phase of inflammation repair. They have been found to suppress tumor-specific T cell activation and proliferation. Presence of TAMS in and around a tumor microenvironment is correlated with a poor prognosis for those suffering from melanoma or breast cancer. Eriksson et al. 2009, looks at
Conclusion
References
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2018, Kenyon College.