Cauliflower mosaic virus
Introduction
By Alan Brennan
The topic of my research is the Cauliflower Mosaic Virus (CaMV). The goal of my research is to identify as well as learn about the structure and function of the Cauliflower Mosaic Virus. Learning of its processes and functions could lead to further studies in transgenic research and increased agricultural efficiency.
Taxonomy
Domain: Virus
- Group: VII
- Family: Caulimoviridae
- Genus: Caulimovirus
- Species: Cauliflower mosaic virus
- Genus: Caulimovirus
- Family: Caulimoviridae
Description and Significance
Mosaic plant diseases are common diseases that occur during warmer months which are caused by plant mosaic viruses. These plant mosaic viruses are identified in plants based on their “mottling” and discoloration of leaves (Britannica). Viruses may also cause leaf deformity, smaller produce and stunted growth. This could lead to plant death and a reduction of crop production which is why the study of mosaic viruses are essential to improve agriculture by both quantity and quality.
Cauliflower Mosaic Virus (CaMV), specifically, effects the Brassicaceae family and impacts a host range including crops such as broccoli, cabbage, cauliflower and turnips.Cauliflower mosaic virus is classified as a Group VII. pararetrovirus. This means the virus requires transcription from its DNA genome to RNA and then reverse transcriptase to transcribe RNA back into DNA genomes in order to produce progeny virions (Slonczewski). It was the first plant virus discovered to utilize a DNA genome and replicate via reverse transcriptase. Cauliflower mosaic virus is usually transmitted from plant to plant via aphid feeding, where virions make their way to insert themselves into the plants nuclear envelope where they inhibit growth and structure of the host.
Cauliflower mosaic virus has been negatively impactful worldwide. Previous studies have shown that CaMV cause a reduction of 25-59% sellable cauliflower heads in Brittany, France as well as a reduction of 60-90% of sellable turnips in the Czech Republic (“Cauliflower Mosaic”). Breakouts of disease can be devastating to agriculture and national economies. Studying and researching this virus could prevent disease and possibly stop the next potato famine of the cauliflower plant.
Cauliflower Mosaic Virus possesses a highly sought after efficient promoter for gene transcription that can be used in an effort to produce transgenic plants (Slonczewski). Transgenic plants are used in order to receive benefits such as producing better yield, quality as well as resistance to insects, diseases and herbicides (Mondal, Sudhadip.).Currently, about 10% of cruciferous vegetables are infected with Cauliflower Mosaic Virus in order to give the host plant pesticide resistance.

Structure and Genome
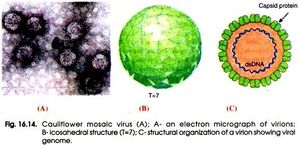
Cauliflower Mosaic Virus is classified as an icosahedral (20 sided) virus. This is because of the icosahedral shape of the capsid that surrounds the viral genome. This shape gives the virus 20 symmetrical triangular faces. This symmetry allows for synthesis efficiency by having the same small number of proteins being produced from a small number genes and from short chromosome sequences. This allows the largest and most complex capsid to be formed from the least amount of resources making it more efficient than a non-symmetrical capsid (Slonczewski). The structure of cauliflower mosaic virus is approximately 52 nanometers in diameter and is surrounded by 420 capsid proteins arranged in triangulation T=7 (P, Ashwathi). In addition to capsid proteins, caulimoviruses are also surrounded by virus associated proteins (“Caulimovirus.”). These proteins are responsible for assisting in the binding of the virus to DNA on its c-terminal end (“InterPro.”).
The efficiency of the icosahedral structure allows it so that the genome of CaMV does not even need to encode for more than 7 genes that already have close similarities. Genes differentiate by where their start codons are so that they have different reading frames (Slonczewski).
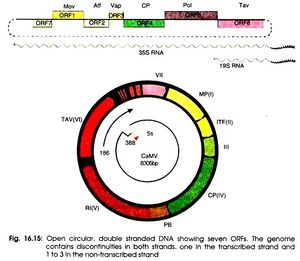
The virion that is encapsulated in the capsid structure is made up of around 8,000 base pairs. This double stranded DNA has an open and circular shape. Also, these strands contain nicks that come from reverse transcription. These nicks are only repaired once these DNA strands enter the host when they form supercoiled molecules responsible for binding to proteins (P, Ashwathi.).
Cauliflower mosaic virus is well known for its strong constitutive 35s promoter. This promoter runs transcription for the entire cauliflower mosaic virus genome. It is because of its efficiency, it is commonly used for studying transgenic plants in relation to gene transfer vectors. The 600 nucleotide leader sequence of the 35s promotor consists of 8 open reading frames that each possess different functions (Padmaratinam). There are 6 major coding regions and 2 minor coding regions. Open reading frame I functions to produce movement proteins to assist progeny viruses to pass through plasmodesmata into uninfected cells (Kobayashi, Kappei.). Open reading frame II produces aphid transmission factors which is required for the transmission of the virus from the aphid to the plant (Leh, V.). Open reading frame III, which previously had no assigned function and is a minor coding region, is believed to promote DNA binding as well as produce structural proteins. ORF IV is responsible for the production of capsid proteins that surround and protect viral genomes. ORF V produces proteins that have proteinases as well as participates in the reverse transcriptase process (Leh, V.). Open reading frame VI produces transactivator proteins that promote formation of inclusion bodies. Inclusion bodies are places of viral multiplication needed to produce many progeny virions. The second minor coding region, open reading frame VII, has an unknown function but is speculated to have some function relating to targeting new viruses and bringing them to inclusion bodies (P, Ashwathi.). A very unique function of open reading frame VI that is encoded by the 19s RNA is that it can reinitiate major reading frames on the 35s RNA when this is usually only found to happen in bacteria (Padmaratinam)

Ecology and Pathology
In order for plant viruses to enter the host’s genome, it must enter via mechanical transmission. This is because cell walls are too thick for some viruses to penetrate so they rely on transmission through broken cells, animal vectors or through their own seeds (Slonczewski). For Cauliflower Mosaic Virus, Aphids are the most common vector for infection. Aphids possess a needle-like mouthpiece that allows penetration into plants. Aphids then ingest the plants nutrients and sap while also leaving saliva behind in the plant. This saliva can infect the plant if the aphid contains a virus coming from a plant that was ingested beforehand (“Aphid-Transmitted Viruses in Vegetable Crops”).
The specific aphid that transports cauliflower mosaic virus is the species Myzus persicae. These fully grown insects grown to be 1.5-2 mm at maturity. They can reproduce at very fast rates and appear during the warmer months to feed on plants. They are polyphageous, which means there is an array of plant species they are able to feed from. Relating to disease, Myzus persicae are known to be able to carry over 100 different types of disease such as cauliflower mosaic virus. Their poisonous saliva can transmit a vast array of diseases from species to species of plant that it chooses to prey on (“Myzus Persicae.”).
Replication
The viral dsDNA genome is introduced via an aphid bite. Once inside the host, the viral genome is un-encapsulated and its DNA is transcribed by the host RNA polymerase to make a copy of viral RNA. Host reverse transcriptase then transcribes copies viral RNA back to DNA. Viral RNA also is used to produce gene products such as new capsid proteins and movement proteins in combination with viral DNA to assemble progeny virions. Capsid proteins house genetic material of viruses and offer protection. Movement proteins interact with plasmodesmata in order to allow for transport that otherwise would not occur (Niehl, Annette). Movement proteins then facilitate movement of progeny virions into uninfected cells of the host plant via plasmodesmata (Slonczewski).
Replication
Conclusion
References
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2018, Kenyon College.
