Colwellia psychrerythraea: Difference between revisions
| Line 47: | Line 47: | ||
==Application to Biotechnology== | ==Application to Biotechnology== | ||
Does this organism produce any useful compounds or enzymes? What are they and how are they used? | Does this organism produce any useful compounds or enzymes? What are they and how are they used? | ||
''C. psychrerythraea''34H, along with many other bacteria that produce polyhydroxyalkanoate (PHA) compounds, as intracellular carbon sources, are of industrial interest for their thermoplastic and elastomeric properties, as well as a source of biodegradable plastic. Similarly, production of cyanophycin-like compounds that serve as nitrogen reserves, also has industrial potential of replacing non-degradable plastics. | |||
==Current Research== | ==Current Research== | ||
Revision as of 02:40, 3 June 2007
A Microbial Biorealm page on the genus Colwellia psychrerythraea
Classification
Higher order taxa
Domain: Bacteria; Phylum: Proteobacteria; Class: Gammaproteobacteria; Order: Alteromonadales; Family: Colwelliaceae; Colwellia
Genus
Colwellia psychrerythraea
Strain: 34H / ATCC BAA-681
|
NCBI: Taxonomy [2] |
Description and significance
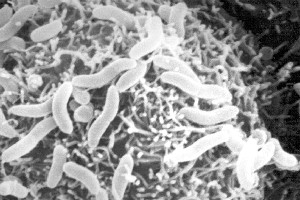
Colwellia psychrerythraea is considered an obligate psychrophile, Gram-negative bacteria, and appears rod-shaped and red in pigment. This flagella-containing organism can be found in continually cold marine environments including Arctic and Antarctic sea ice. Strain 34H, in particular, was isolated from Arctic marine sediments. It has a growth temperature range of -1°C to 10°C. Optimal growth appears at 8°C, with maximum cell yield occurring at the subzero temperature of -1°C. Cells are able to swim in temperatures as low as -10°C. Growth can occur under deep sea pressures as well.
Sequence completion of this genome has revealed Colwellia's significant role in bioremediation, carbon and nutrient cycling, production of secondary metabolites, and cold-adapted enzymes. C. psychrerythraea is considered a model organism for the study of life in permanently cold environments, specifically bacterial adaptations. These adaptations include production of extracellular polymeric material for purposes of biofilm formation and cryoprotection, as well as enzymes with the ability to breakdown high-molecular-weight organic compounds. Particularly unique to this organism is the production of cold-active enzymes which show distinct heat instability and optimal activity occurring at low temperatures. These features make Colwellia species important in carbon and nutrient cycling in the cold marine environments in which they inhabit. From contaminated sediments to ice formations, observation of this organism can possibly give insight into earlier Earth environments as well as those on other planets and moons.
Genome structure
The complete genome of Colwellia psychrerythraea 34H has been sequenced and shown to be 5,373,180 nucleotides in length. The genome consists of one circular chromosome which codes for 117 structural RNAs and 4910 proteins. The circular DNA contains 38% guanine-cytosine base pairing.
Comparative genome analyses proposes that the psychrophilic behavior stems from a set of synergistic modifications in the overall genome content and amino acid composition, rather than a specific collection of genes responsible for such cold adaption.
Cell structure and metabolism
This rod-shaped, Gram-negative bacteria is enclosed by two cell membranes and contains flagella for motility. Important adaptations to its survival in such cold environments include production of extracellular polysaccharides which function in protection against cold-damage, known as cryoprotection, as well as a large amount of extracellular enzymes which may play a role in the metabolism of dissolved organic carbon at low temperatures.
Other cell features characteristic to cold adaption involve cell membrane fluidity and coding sequences for polyunsaturated fatty acid synthesis as well as a fatty acid cis/trans isomerase, which both aide in increasing cell membrane fluidity.
Genomic investigations also show C. psychrerythraea produces polyhydroxyalkanoate (PHA) compounds, which function in intracellular carbon and energy storage. Means of nitrogen storage has also been identified in the production cyanophycin-like compounds, which serve as nitrogen reserves. Production of both of these compounds insures important intracellular carbon and nitrogen reserves when cold environments may set limitations on such uptake.
Ecology
C. psychrerythrea's unique cold adaptations such as the ability to breakdown high molecular weight compounds and carbon metabolism make this organism important in carbon and nutrient cycling in such cold marine environments. Detoxification activities by this organism may aide in bioremediation of cold habitats as well.
Pathology
This organism is not known to be pathogenic.
Application to Biotechnology
Does this organism produce any useful compounds or enzymes? What are they and how are they used?
C. psychrerythraea34H, along with many other bacteria that produce polyhydroxyalkanoate (PHA) compounds, as intracellular carbon sources, are of industrial interest for their thermoplastic and elastomeric properties, as well as a source of biodegradable plastic. Similarly, production of cyanophycin-like compounds that serve as nitrogen reserves, also has industrial potential of replacing non-degradable plastics.
Current Research
As of May 2007, scientists at the Department of Biomedicine at the University of Bergen, has solved the crystal structure for the cold-adapted enzyme phenylalanine hydroxylase (CpPAH) found in C. psychrerythraea 34H. With the aide of that structure, they analyzed the catalytic properties of the enzyme, and found that the cold-adaptive properties appear to involve increased flexibility around the active-site of CpPAH as well as hydrogen bond disruption for the enzyme's cofactor BH4. CpPAH exhibited highest activity at 25(o)C, which is 15(o)C above the optimal temperature growth. Characteristic of cold-active enzymes, high catalytic efficiency of the enzyme was observed at 10(o)C. However, unlike most cold-adapted enzymes, the half-inactivation and denaturation, known as the Tm, was found to be only slightly lower when compared to this enzyme's activity in non-psychrophilic organisms. This knowledge, along with observance of the crystal structure, suggests that cold-adaption for CpPAH involves increased flexibility, which do not largely effect the enzyme's thermostability.
Researchers in Toulouse, France analyzed TonB-dependent receptors (TBDRs), which are outer membrane proteins primarily recognized for the uptake of iron siderophore complexes in Gram-negative bacteria, and their role in Proetobacteria, as well as Bacteroidetes. Genome analysis has identified large representation of these TBDRs, in both phytopathogenic, and aquatic bacteria including C. psychrerythraea. Genomic analysis of both types of bacteria show the TBDRs include a membrane transporter, an amylosucrase which aides in sucrose hydrolysis, and a sucrose utilization regulator; these make up a newly identified carbohydrate utilization locus, named CUT locus. These specific TBDRs and CUT loci have been found to be different from TBDRs in human gut Bacteroids, which allow for glycan scavenging. This suggests a convergent evolution relationship between TBDRs in Proteobacteria and Bacteroidetes.
Scientists at the University of Washington, which have performed extensive research on C. psychrerythraea 34H, have discovered a new temperature minimum for metabolic activity in this organism, which involves incorporation of leucine into proteins at the low temperature of -20(o)C. Effects of added extracellular polymeric substances (EPS), which can aide in attachment, were also analyzed and found to enhance rates of leucine incorporation in temperatures -1 to -20(o)C. Unexpectedly, low rates of intracellular incorporation of leucine into protein were observed in samples with added EPS, at the low temperatures of -80 and -196(o)C. Explanation of such mechanisms is unclear but may involve salt and organic polymeric substances. Further analysis of bacterial extracellular-intracellular relationships could aide in the understanding of activity at sub-freezing temperatures.
References
|
NCBI: Colwellia psychrerythraea 34H Genome Project at TIGR [3] |
Edited by Jaclyn Gaede; student of Rachel Larsen and Kit Pogliano
