Life on Mars: Difference between revisions
SteigmeyerA (talk | contribs) |
No edit summary |
||
| Line 1: | Line 1: | ||
{{Curated}} | |||
==Introduction== | ==Introduction== | ||
[[Image:Angryred.jpg|thumb|300px|right|Fig. 1 The "Bat-rat-spider-crab" from the 1959 science fiction film "The Angry Red Planet" is a highly unlikely form of Martian life. American International Pictures 1959.]] | [[Image:Angryred.jpg|thumb|300px|right|Fig. 1 The "Bat-rat-spider-crab" from the 1959 science fiction film "The Angry Red Planet" is a highly unlikely form of Martian life. American International Pictures 1959.]] | ||
Latest revision as of 15:06, 13 July 2011
Introduction
While observing Mars during the planet’s “Great Opposition” in 1877, Italian astronomer Giovanni Schiaparelli saw dark lines crisscrossing the surface of the planet. He reported them as “canli,” the Italian word for channels (AMNH). This finding was misleadingly translated into English as “canals” – a word that has a certain “suggestion of intelligent design,” according to astronomer Carl Sagan (Cosmos). This event triggered a worldwide fascination with the Red Planet and speculation on what life may exist there.
At the turn of the 20th Century, H.G. Wells portrayed a violent race of advanced Martians regarding the Earth with envious eyes and leaving their dying world to conquer ours with their great machines. Around the same time, Edgar Rice Burroughs wrote of John Carter, a gentleman from Virginia who traveled to “Barsoom,” the native’s name for Mars – a dying planet divided by two warring races: the red humanoids of Helium and the green, four-armed giants known as the Tharks.
The Martians of 1950s b-sci-fi films became even more outlandish. Creatures like the stocky, brown, three-eyed invaders in the 1953 adaptation of The War of the Worlds or the giant “bat-rat-spider-crab” from 1959’s The Angry Red Planet (Fig. 1) scurried across the blood-red soils of a fictional Mars. Though excitement for this alien world was building, our first glimpses of this mysterious neighbor, in the decades to come, would be somewhat anti-climactic.
The Mariner 3 spacecraft gave humans the first close-up look at Mars during a flyby in 1964. The Viking landers sent back the first pictures from the surface of another planet in 1976. The images from these probes showed a world that seemed almost familiar. The vast rocky planes and crooked mountain peaks closely resembled Earth deserts. If not for the rust-colored soil, Mars may not have seemed alien at all. Undoubtedly many people were disappointed that our probes did not capture a little green man waving back at the camera. But, even with those first missions, evidence began to mount for the presence of small, but certainly not insignificant, Martian life.
Recent evidence nearly conclusively proves that Mars was once a much warmer and wetter planet. Conditions on Mars could have been very conducive to life several billion years ago. Unlike Earth, however, Mars failed to maintain an atmosphere and the liquid water evaporated into space and the planet became increasingly arid and vulnerable to high levels of radiation. If life existed before this climate change, could it have evolved fast enough to survive? Even with the planet’s extreme conditions, could there be small oases capable of supporting microbial life? It wouldn’t take a six-foot, bug-eyed beast with eight tentacles to profoundly alter our views of the universe – all we need is just one microscopic cell.
Notable Mars Missions and Findings
Viking 1 and 2 — Reached Martian surface on July 20, 1976 and September 1976, respectively. These landers were designed to analyze the Martian soil, atmosphere composition, weather and geographic features. The labeled-release experiment found that organic material was consumed when exposed to a soil sample, as if life were present. This contradicted a gas-chromatography experiment that yielded no evidence of organic compounds in the soil (Navarro-Gonzalez 2003). However, recent evidence suggests that these may have been low levels of organic compounds that the experiments were not sensitive enough to analyze, or that the organic compounds were too stable to be converted into a gas and thus were undetectable in the spectral analysis. (Handwerk 2006).
Mars Global Surveyor — Reached Mars orbit on September 12, 1997 and was operational through 2006. This orbiter was designed for extensive mapping of Mars and the study of daily weather patterns. It recorded images of gullies, debris flows and other evidence of surface water in Mars’ distant past as well as CO2 ice, which is slowly receding at the poles (NASA JPL).
Mars Pathfinder — The first successful lander since the Viking program, the rover Sojourner was designed to analyze the geological history of Mars, determine soil composition and look for signs of life. The landing site (Fig. 4) was selected because scientists believed that the area had once been the subjected to a large flood. The mission provided evidence suggesting that Mars had a warmer and wetter climate in the past (NASA JPL).
Mars Odyssey — The orbiter arrived at Mars on October 24, 2001 and conducted a planet-wide geological survey mission. It successfully mapped out mineral deposits across the planet and identified areas of water-ice just below the surface. The probe also determined that radiation in low-Mars orbit is twice that in low-Earth orbit (NASA JPL).
Spirit and Opportunity Rovers — After landing on Mars on June 10, 2003 and January 24, 2004, respectively, these rovers began long distance and long duration exploration of the planet. They were far more mobile than previous rovers and contained tools to examine the interiors of rocks. Opportunity discovered a dry lakebed and found many sulfate deposits in the sedimentary bedrock. The sulfates have since been interpreted as chemical deposition by evaporative processes or alteration due to groundwater circulation (Stivaletta et al. 2009), providing strong evidence for past surface water. Spirit examined a basaltic region, shaped greatly by volcanic activity (Fig. 3) (NASA JPL).
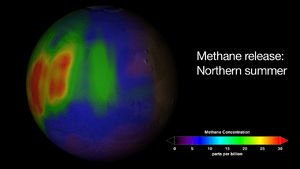
Mars Express — This probe, which reached Mars in 2004, consists of an orbiter and a lander, both designed to look for evidence of water beneath the surface and examine evaporite deposits. The orbiter discovered that 15% of South Pole ice is water and the rest is CO2. Analysis of the atmosphere showed a continually renewed concentration of methane (Fig. 2). The source of this gas is unclear but, since it disappears quickly from the air (possibly due to hydrogen-peroxide in the soil (Gough et al. 2011), it must be replenished regularly. Methane-producing microbial life is one possible source (NASA JPL), although it could be the result of natural geologic processes, like iron oxidation (Steigerwald 2009).
Mars Reconnaissance Orbiter — Since March 2006, this orbiter has been taking the most detailed and close-up pictures of Mars ever captured from orbit (Fig. 5). The goal is to study the Martian soil and build on NASA’s geographic and sediment maps of the planet. It also contains a new communication system known as “Interplanetary Internet,” which allows for faster and more direct links with Mission Control and with future spacecraft.
Phoenix — The lander descended to the Martian Polar region under its own power on May 25, 2008. Instruments detected chloride, bicarbonate, magnesium, sodium potassium, calcium, and traces of sulfate in the soil. The most important discovery was the presence of Perchlorate, a strong oxidizer in the soil that could contribute to biological metabolism as an energy source for anaerobic reduction and through its hygroscopic properties. This chemical could also be used for rocket fuel or as a source of oxygen for future manned missions (NASA JPL).
Current Mars Conditions
Arid Environment — Mars has an atmosphere much thinner than Earth’s. As the atmosphere began to disappear in Mars’ distant past, surface and atmospheric water evaporated into space. Probes have detected liquid and frozen water just below Mars’ surface, at the polar ice caps and in the form of early morning fogs. Soil water content increases with latitude from 2-5 wt.% in low-latitude regions to 5-15 wt.% approaching the poles (Marlow et al. 2010). Desiccation most negatively affected microbes exposed to an arid laboratory environment, analogous to Mars, – more so than radiation, high salinity or low pressure (Berry et al. 2010).
Radiation— The UV radiation penetrating the Martian atmosphere is three orders of magnitude greater than on Earth and poses one of the most significant threats to microbes exposed to the air (Marlow 2010). Mars’ atmosphere does not absorb UV radiation so levels near the surface fluctuate significantly (Marlow 2010). Mars surface levels of UV radiation prevented populations of E. coli from growing or reproducing as they were essentially sterilized (Berry et al. 2010). One theory for the inconclusive results of the organic molecule tests conducted on the Viking landers is the presence of this sterilizing radiation that degrades organic molecules once they are removed from the soil (Stockton et al. 2010). Microbes cannot survive long on the Martian surface.
This radiation has been shown to cause cells to quickly denature. Schuerger et al. created a Mars simulator and subjected populations of B. subtilis to Martian surface levels of radiation. Within 15 seconds, there was a 99% loss in cell viability (Schuerger et al. 2003). A similar experiment conducted with the endolithic cyanobacterium Chroococcidiopsis sp. saw a 99% loss in cell viability after 5 minutes and the death of all cells after 30 minutes (Cockell et al. 2005). However, even after the loss of viability, enzymatic activity and pigmentation, cell DNA was still detectable, suggesting that evidence of biological processes can persist, even under this intense radiation (Cockell et al. 2005). This may be an important factor in the detection of biological signatures in the Martian soil.
Martian Seasons — Mars has a tilt of 23.5°, which is very similar to Earth’s 25.2° tilt. The two planets therefore share similar seasonal patterns. However, Mars has a longer solar orbit so its seasons last twice as long as Earth seasons (James 2011). When Mars is closest to the sun, it can reach temperatures as high as 27°C, which result in dust storms that absorb sunlight and can further heat the planet (Wikipedia). In the winter, the polar ice caps can reach temperatures as low at -143°C, while common temperatures across the planet remain around -55°C.
Possible Methods of Microbial Survival
Finding Water in a Dry Habitat
The robotic survey missions have provided evidence of a much warmer and wetter climate in Mars’ distant past. The Mars Exploration Rover “Opportunity” detected sedimentary evaporates in salt-water “sabkhas” – salt flats that normally form on arid shorelines (Golombek et al. 2005). Orbiters have detected further evidence of a salt-water history in the saline brine pockets and evaporite deposits found on the surface (Crumpler et al. 2005; Wierzchos et al. 2011). Some of the water flowed in small streams or rivers, as evidenced by the many carved features of the Martian landscape (Stivaletta et al. 2009) while other bodies of water existed planet-wide in extensive reservoirs, but never in large enough quantity to be classified as oceans, based on distributions of evaporite deposits (Osterloo et al. 2008). Though some areas of water lasted longer, most surface water dried up around 3.7 billion years ago (Crumpler et al. 2005). Today, is there any water remaining on Mars and is it enough to sustain life?
When Mars’ atmosphere deteriorated and the planet cooled, much of the water on the surface and in the atmosphere evaporated into space. However, there is strong evidence to suggest that large amounts of water were absorbed into the soil and remain to this day in the form of ice. The Phoenix Lander uncovered ice just below the surface and the Mars Reconnaissance Orbiter detected a white material, which appears in craters following meteorite impact and fades over time (Marlow et al.). Though it may be possible for organisms to use the water-ice, a prominent source of water could be in the supposedly low-water atmosphere. Just before and shortly after sunrise, the moisture level on Mars is close to saturation. The Phoenix Lander observed ground fogs forming and drops of liquid water condensed on the lander’s legs (Fig. 6) (Schulze-Makuch 2010). Environments with high relative humidity (RH) can trigger the metabolic activity of phototrophic organisms, even without the condensation of water. The cryptoendolithic lichens in Death Valley sandstone, for example, will begin photosynthetic processes at RH greater than 70% (Wierzchos et al. 2010). On Mars, though, the key to xerophilic organisms’ ability to utilize water vapor may lie in the properties of Perchlorate.
Perchlorates are powerful oxidizers, can work as anti-freeze agents and are very hygroscopic (Schulze-Makuch 2010). The Phoenix Lander detected high concentrations of this chemical in the Martian soil (Marlow et al. 2010). Martian microbes could incorporate Perchlorates into their metabolism and use it to obtain water from the atmosphere. On Earth, Perchlorate-reducing bacteria exist in anoxic environments by oxidizing organic carbon or inorganic donors like H2, H2S or Fe2+ and couple that oxidation to Perchlorate reduction (Schulze-Makuch 2010). On Mars, Xerophilic microbes could use the antifreeze properties of Perchlorate to survive the cold temperatures, the hygroscopic properties to pull water from the early morning mists and the oxidizer properties for chemical processes. Earth organisms with these adaptations are usually halophilic, favoring salt water as an internal solvent (Schulze-Makuch 2010). Perhaps not coincidentally, many of the evaporite deposits suggest a high-salt content in the Martian soil.
Salinity on Mars
Using data from the Thermal Emission Imaging System on the Mars Odyssey and supporting data from the orbiting probes Mars Global Surveyor and Mars Reconnaissance Orbiter, Osterloo et al detected the presence of chloride salts on Mars (Fig. 7) These saline minerals were deposited in the soil either after weathering of basaltic rocks or through erosion created by ancient lakes and rivers. As these bodies evaporated, the salts precipitated from the solution and left deposits spotted across the planet (Osterloo et al. 2008; Stivaletta et al. 2009). When exposed to this level of salinity in a laboratory Mars-analogue environment, E. coli were able to survive, but there was no increase in their density or population size (Berry et al. 2010). It is therefore feasible that organisms could live in the Martian soil if they are properly adapted.
The presence of these deposits, and their similarity to the sabkhas on Earth, have made halophilic organisms a likely candidate for life on Mars (Stivaletta et al. 2009). On Earth, prokaryotic inhabitants of sabkhas (salt flats) accumulate KCl in their cells to maintain osmotic similarity to the surrounding soil (Stivaletta et al. 2009). But severe osmotic difference with the environment is not the only threat that these organisms have adapted to on Earth. Halophiles living in sabkhas face drastic temperature changes and severely dry conditions. To solve these problems, the organisms seek shelter in the gypsum crystals of halite crusts — prominent features of the Martian surface.
Halite and Gypsum Shelters
The Atacama Desert of Chile is considered to be a close analogue of Mars due to its low average precipitation (less that 1m per year) and 3-4 million years of persistent hyperarid conditions (Wierzchos et al. 2010). The basic soils in this desert harbor little to no life but the halite crusts, which contain microporous gypsum crystals, are abounding with photosynthetic microorganisms, suggesting that it was sought as a refuge once the area dried millions of years ago. This microhabitat supports organisms across different phyla including cyanobacteria and proto-bacterial lineages (Wierzchos et al. 2010).
Gypsum has many features that make it a haven for extremophiles. Gypsum is a very hygroscopic molecule and masses of gypsum microcrystals that condense liquid at low RH will retain that water, thus offering oases in arid environments (Wierzchos et al. 2010). In addition to being a harbor against desiccation, gypsum is a substrate that can limit exposure to UV radiation (Marlow et al. 2010), while, at the same type, having enough transparency to allow for sub-surface photosynthesis (Stivaletta et al. 2009, Wierzchos et al. 2010). Phototrophic cyanobacteria are common inhabitants of the pore spaces between gypsum crystals because they can exist in a less-exposed, wetter, sub-surface environment, but still conduct essential photosynthetic processes. In extremely arid regions, as in the Atacama desert, photochemical processes dominate (Navarro-Gonzalez et al. 2003). The creation of mineral solutions in the crusts give organisms much better access to minerals and a higher moisture content than direct exposure on the surface (Stivaletta et al. 2009). Halite has been found in Martian meteorites (Stivaletta et al. 2009) and there are widespread deposits of gypsum in the Olympia Undae sand sea near the polar ice cap (Wierzchos et al. 2010) — an area that is known to be heavy in mineral and water deposits.
Gypsum is a sulfate with a comparatively low solubility so it will quickly precipitate from evaporating water (Fishbaugh et al. 2007). The Gypsum deposits discovered in the Juventae Chasma of Mars were most likely the result of sulfur-bea ring water interacting with Ca-bearing minerals, leaving gypsum behind as it evaporated, based on the findings of the Opportunity rover (Fishbaugh et al. 2007). Gypsum can also be formed as sulfates and other salts accumulate when water is not present to remove them from the environment. Changes in humidity can hydrate anhydrate, transforming it into gypsum (Wierzchos et al. 2010). The only positive identification of these types of deposits has occurred in the north polar sand seas (Fishbaugh et al. 2007). Adding to the feasibility of this location as a prime habitat for life, the Mariner probes demonstrated that soil water content increases with latitude from 2-5 wt.% in low-latitude regions to 5-15 wt.% approaching the poles (Marlow et al. 2010). It is conceivable that as water disappeared from the Martian soil and atmosphere, organisms sought refuge in these halite crusts and may continue to exist there.
Possible Types of Modern Martian Life
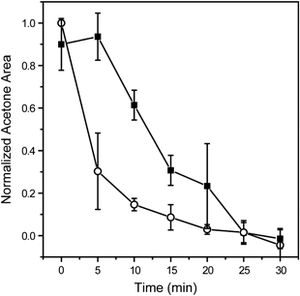
It is possible that some form of life existed on Mars in the past with its thick atmosphere, warmer temperatures, mineral deposits and abundant water. Even if extremophiles were able to survive the planet’s dramatic climate changes, were they able to evolve fast enough to exist on modern-day Mars? Extra-terrestrial oxidized organic molecules, like aldehydes and keytones, have been detected in the Murchison meteorite (Stockton et al. 2010) so it is likely that evidence of organic metabolic processes could be found on Mars. The first probes on Mars may have detected evidence of biochemical processes but their sensors were not sensitive enough to give definitive results (Handwerk 2006). Recently, the Mars Organic Analyzer on the Phoneix Lander successfully detected acetone in a Mars soil sample and found that these types of molecules have a short lifespan when exposed to the atmosphere (Fig. 8) (Stockton et al. 2010, Johnson et al. 2011). This not only offers a possible explanation for why the Viking landers’ results were inconclusive, but, more importantly, shows that organic molecules cannot survive on the surface, but do exist under the soil. If living microorganisms exist under the soil, what form would they take?
Martian Extremophiles
Mars Express and the Mars Reconnaissance Orbiter have uncovered extensive deposits of concentrated sulphate salts, which suggest that Martian water was 10-100 times more saline than Earth seawater so if microorganisms exist there today, they would most likely be halophiles (Marlow et al. 2010). On Earth, most bacteria live in environments with an availability of water (aw) between 0.9 and 1.0, but some organisms, like Xeromyces fungi can exist in an aw as low as 0.6 (Marlow et al. 2010). Mars aw values at Meridani Planum range between 0.51 and 0.78 so it is conceivable that Earth-like halophiles could exists in that region. However, extreme halophiles on Earth that have adapted to these conditions, evolved from less tolerant organisms (Marlow et al. 2010) so these salt brines may not be able to support the formation of life. The Meridani Planum region has been osmotically challenging since the Noachian period (between 3 and 4 billion years ago) so it may have been difficult for life to arise there at all (Marlow et al. 2010).
Another challenging aspect of these saline regions is their high acidity. The rover Opportunity detected jarosite, magnesium sulphates and calcium sulfates in this soil, suggesting not only a high concentration of salts, but also a high acidity (Stockton et al. 2010; Marlow et al. 2010). This would require some acidophilic mechanisms in any organisms living there. Some acidophiles will pump protons out of the intracellular space to maintain a neutral pH, while some organisms, like the Acetobacter aceti, utilize acid-stable proteins that prevent an over accumulation of cations (Marlow et al. 2010). There is precedent for extreme acidophiles like the achaea species Picrophilus oshimae that lives at a pH of 0.7.
Though the northern halite crusts may offer protection from dessication and radiation (as long as the organisms are halophilic and acidophilic), they will not protect microbes from the harsh temperature fluctuations that occur across the planet. The Mars Global Surveyor observed Martian weather patterns for a year and recorded temperatures between -125°C and 25°C. Below the surface, the temperature would most likely be below the freezing point of water year-round (Marlow et al. 2010). However, there are instances where organisms can survive in nearly perpetually frozen environments. In Antarctica, the sun will cause localized melts in the ice for 150 days out of the year. These melted microhabitats are nutrient-rich solutions that become oases for microbes, including cyanobacteria (Priscu et al. 1998). In the nutrient-rich soils of Mars, it is conceivable that some pockets of frozen water could become liquefied long enough to support life. This is even more likely within the gypsum deposits, due to the mineral’s anti-freezing properties.
Conclusions
If life is present on Mars, it will most likely be in microbial form. Living beneath the underground seems to be the only solution to the extreme desiccation, radiation and temperature effects on the surface. Based on the evidence shown here, the area of focus for future missions should be the saline deposits in regions just south of the North Pole. Not only are the soils in these regions rich in minerals, they also contain the highest percentage of water. The gypsum deposits in the dry lake and river beds could offer shelters from radiation and extreme temperature fluctuations, while allowing subsurface photosynthesis to occur. Gypsum and perchlorates could help pull gaseous water from the early morning mists and condense it in micropores within the soil. Microbes could thrive in the saline solutions formed in these halite-crust microhabitats.
Even with all the advantages of the dry waterways in the North, the organisms would still have to be adapted to several extreme conditions. The water in the soil micropores would be saline and extremely acidic. Although organisms exist in both types of environments on Earth (well within the range of Mars conditions), Martian microbes would have to be multi-faceted extremophiles. They would have to incorporate traits of halophiles, acidophiles, thermophiles, xerophiles and utilize anoxic and photosynthetic metabolism. It is not difficult finding an organism that exists under a couple of these environmental stresses, but can there be life so precisely evolved that it can survive in a highly specific niche? The answer can only come with time.
The early 21st Century has seen an unprecedented level of Mars exploration. Dozens of orbiters, robots and scientific instruments have been combing the planet for over a decade and we have learned more about Mars in that short time than in all of human history. Evidence for life on the Red Planet, past and/or present, is mounting. All we have to do is keep looking for this century’s little green men.
— August Steigmeyer
References
Wikipedia. “Climate of Mars.” Wikipedia.org, April 18, 2011.
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2009, Kenyon College.