Nipah Virus
INTRODUCTION
At right is a sample image insertion. It works for any image uploaded anywhere to MicrobeWiki. The insertion code consists of:
Double brackets: [[
Filename: PHIL_1181_lores.jpg
Thumbnail status: |thumb|
Pixel size: |300px|
Placement on page: |right|
Legend/credit: Electron micrograph of the Ebola Zaire virus. This was the first photo ever taken of the virus, on 10/13/1976. By Dr. F.A. Murphy, now at U.C. Davis, then at the CDC.
Closed double brackets: ]]
Other examples:
Bold
Italic
Subscript: H2O
Superscript: Fe3+
FUN FACT
The Nipah virus was the virus which inspired the MEV-1 virus in the 2011 Hollywood hit “Contagion”.
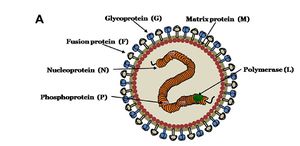
MORPHOLOGY OF NiV
Nipah virus is in the newly created Henipavirus genus with the closely related Hendra virus and Cedar virus. The Henipavirus family is pleomorphic, meaning their shape is varied, and traditionally 40 to 600 nm in diameter. The core of a virion contains a linear ribonucleprotein (RNP) comprising of negative sense single stranded RNA. Also present in the RNP are three critically important proteins. Nucelocapsid proteins (N) are tighly bound to the various nucleotides of the RNA strand. N protein is the most abundant protein present and necessary for capsid structure. Phosphoproteins (P) and large polymerase proteins (L) are also bound to the RNA and aid RNA polymerase in transcribing RNA to mRNA to antigenomic RNA. The virion is enveloped by a traditional lipid bilayer but “spiked” with fusion (F) and receptor-binding glycoproteins (G). The fusion proteins are responsible for fusing the viral membrane to the host membrane triggering the release of the contents of the virion. The receptor-binding glycoporteins are extremely specific and bind only to Ephrin B2 (EFNB2) surface proteins. Specifically, NiV has been found to alternatively bind to EFB3 as well. The EFNB2 surface proteins are highly conserved across the mammalian lineage. On the underside of the lipid bilayer matrix proteins (M) are present for structural support and regulating the budding process. Other proteins, C, V, and W, are also present in the cytoplasm and involved in regulation of transcription and replication.
In regards to the Nipah virus genome, the exact structure is not completely understood. However because of the strong homology between Hendra virus and Nipah virus, a nearly identical structure is hypothesized. The negative sense single stranded RNA is of traditional 3’ to 5’ orientation. All the previously mentioned proteins are encoded by the RNA in the order of 3’-N-P-M-F-G-L- 5’. Similar to all paramyxoviruses NiV RNA replication occurs in the cyptoplasm. All but the P gene are monocystronic, in that they code for a single protein. The P gene also encoded for the C, V, and W proteins which play a role in the virulence of NiV.
Interferons are released by host cells when under attack by a pathogen which enables intercellular communication. The intercellular communication is necessary for the triggering of immune cells which get rid of the pathogen. C, V, and W proteins, encoded by the P gene, have anti-interferon activity in that they block the transcription of interferon signaling. The process by which C, V, and W proteins block the signaling is still unknown.
NiV OUTBREAK HISTORY
Include some current research in each topic, with at least one figure showing data.
OVERVIEW OF NiV TRANSMISSION
MALAYSIA/SINGAPORE OUTBREAK TRANSMISSION OF NiV
BANGLADESH/INDIA OUTBREAK TRANSMISSION OF NiV
REPRODUCTION OF PARAMYXOVIRUSES
PATHOLOGY
DETECTION AND TREATMENT OF NiV
CONCLUSION: THE FUTURE OF NiV
Overall paper length should be 3,000 words, with at least 3 figures.
References
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2009, Kenyon College.