Symbiodinium Symbiosis
Characteristics of the Symbiont: Symbiodinium sp.
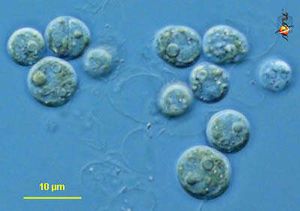
Phylogeny: Domain Eukaryota, Kingdom Chromalveolata, Phylum Dinoflagellata, Class Dinophyceae, Order Suessiales, Family Blastodiniaceae, Genus Symbiodinium
Symbiodinium is a genus of diverse endosymbiotic algae with genus members commonly referred to as zooxanthellae (Blank 1987, van Oppen et al. 2001, and Weis et al. 2001). Being algae, organisms of symbiodinium are autotrophic and eukaryotic, gaining energy from photosynthesis (Blank 1987). Individual cells are coccoid and at times produce flagella to enhance motility, making them dinoflagellates (Blank 1987). Symbiodinium sp. have the smallest genomes of all known dinoflagellates, but at 3pg of DNA per cell the genome is even larger than that of humans and has not been completely sequenced yet (LaJeunesse et al., 2005; and Lin, 2006).
Characteristics of the Host: Coral

Phylogeny: Domain Eukaryota, Kingdom Animalia, Phylum Cnidaria, Class Anthozoa
Many coral species form obligate endosymbiotic relationships with dinoflagellates (van Oppen et al. 2001). For example, the Hawaiian stony coral (Montipora verrucosa), corals of the genus Acropora, and Fungia scutaria all rely on zooxanthellae for survival (Blank 1987, van Oppen et al. 2001, and Weis et al 2001).
Host-Symbiont Interaction
Zooxanthellae are found in gastrodermal cell vacuoles of the coral they reside in (Weis et al. 2001).
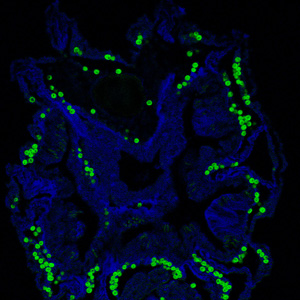
Gastrodermal cells are cells that line the gastrovascular cavity, where nutrients are digested and distributed (Wikipedia). Coral are born azooxanthellate, symbiont free, and are infected with the symbiont horizontally while in the polyp stage through either feeding or phagocytosis by gastrodermal cells (Weis et al. 2001). Zooxanthellae provide photosynthetically fixed nitrogen to coral while coral provides inorganic nutrients, a high light environment, and protection from the threat of aquatic herbivores (Weis et al. 2001). Without zooxanthellae coral experience severely reduced growth, survivorship, and fitness (Weis et al. 2001). Coral form highly specific relationships with zooxanthellae, only taking in one zooxanthellae species per colony. The most successful coral-zooxanthellae relationships occur with homologous strains (Weis et al. 2001). Coral infected with zooxanthellae only found in other coral species either form a weak symbiotic relationship or lose the symbiont altogether (Weis et al. 2001).
Molecular Insights into the Symbiosis
Available data are still insufficient to describe the complete molecular systematics of the exchange of nutrients between Symbiodinium sp. and corals (Carlos et al., 1999).
However, the study of coral bleaching, or a coral's loss of its symbiont, gives insight to the specific pathways coral and symbiodinium interact. Corals are considered 'bleached' because the loss of algal symbionts lead to loss of pigmentation, and the color left is that of the underlying skeleton. Coral bleaching occurs during periods of exposure to elevated water temperatures and can be affected by a variety environmental factors (Warner et al., 1999). One contributing factor to dinoflagellate loss is damage to photosystem II, in particular the decline in number of D1 reaction center proteins (Warner et al., 1999). With damage to a protein important to maintaining photosynthetic function, these damaged and lost symbionts have a reduced, if not absent, photosynthesis levels. It is hypothesized that host corals expel the dysfunctional symbionts because they no longer produce fixed nitrogen (Warner et al., 1999).
A recent study on the scleractinian coral Ctenactis (Fungia) echinata found evidence that coral lectins, sugar-binding proteins, play a role in engaging zooxanthellae and maintaining symbiosis (Jimbo et al., 2010). Since corals are known to be species specific with their symbionts and can expel symbionts, there is some kind of recognition ocurring (Jimbo et al., 2010). Recognition occurs during the first few hours of the onset of infection through a lectin/glycan interaction (Jimbo et al., 2010). In Jimbo et al.'s (2010) experiment lectins transformed flagellate Symbiodinium cells (a motile form) into the coccoid form taken on during symbiosis. Lectin interaction could also be involved in the loss of Symbiodinium cells (Jimbo et al., 2010).
Ecological and Evolutionary Aspects
The relationship between host and symbiont genotype is unclear and no evidence of coevolution has been found, however there are light related patterns with distribution (van Oppen et al., 2001). In one host colony observed by van Oppen et al. (2001) there were different symbiont clades in shady areas versus those exposed to light. It appears that the occurrence of Symbiodinium taxon in coral species depends on a combination of factors including symbiont availability, ambient light levels, host genetic identity, and competition between zooxanthella taxa (van Oppen et al., 2001).
Recent Discoveries
Host/Symbiont Recognition Mechanisms: Vidal-Dupiol et al. (2009) found two coral genes coding for proteins Pdcyst-rich and PdC-Lectin, which are involved in maintaining skeletal health and the zooxanthellae symbiosis. The decrease in Pdcyst-rich transcripts was found to correlate with a decrease in skeletal calcification, displaying its involvement with mineralization (Vidal-Dupiol et al., 2009). PdC-Lectin was found to have the ability to bind to Symbiodinium cells, and its down regulation correlated with low levels of zooxanthellae cells (Vidal-Dupiol et al., 2009). Because these two genes are down regulated at the onset of bleaching, Vidal-Dupiol et al. (2009) hypothesized that bleaching occurs partly because of a decrease in zooxanthellae aquisition.
Radical Formation Undermining Symbiosis: Higuchi et al. (2010) found that hydroxyl radicals, the most toxic reactive oxygen species, form in coral and zooxanthellae tissues under high levels of environmental stress, such as high levels of UV radiation and high temperatures. This production of hydroxyl radicals causes oxidative damage to organelles and photosystem II, reducing photosynthesis levels and (Higuchi et al., 2010). Thus, giving more insight to the complex pathways leading to coral bleaching, which is thought to be an indicator of environmental health and climate change.
References
Blank, R.J. 1987. Cell architecture of the dinoflagellate Symbiodinium sp. inhabiting the Hawaiian stony coral Montipora verrucosa. Marine Biology 94: 143-155)
Carlos, A., Baillie, B., Kawachi, M., and Maruyama, T. 1999. Phylogenetic position of symbiodinium (dinophyceae) isolates from tridacnids (bivalvia), cardiids (bivalvia), a sponge (porifera), a soft coral (anthozoa), and a free-living strain. Journal of Phycology 35: 1054-1062.
[1] Higuchi, T., Fujimura, H., Hitomi, Y., Arakaki, T., Oomori, T., and Suzuki, Y. 2010. Photochemical formation of hydroxyl radicals in tissue extracts of the coral Galaxea fascicularis. Photochemistry and Photobiology 86: 1421-1426.
[2] Jimbo, M., Yamashita, H., Koike, K., Sakai, R., and Kamiya, H. 2010. Effects of lectin in the scleractinian coral Ctenactis echinata on symbiotic zooxanthellae. Fisheries Science 76: 355-363.
[3] LaJeunesse, T., Lambert, G., Anderson, R., Coffroth, M. A., and Galbraith, D. 2005. Symbiodinium (pyrrhophyta) genome sizes (DNA content) are smallest among dinoflagellates. Journal of Phycology 41: 880-886.
[4] Lin, S. 2006. The smallest dinoflagellate genome is yet to be found: a comment on LaJeuness et al. "Symbiodinium (pyrrhophyta) genome sizes (DNA content) are smallest among dinoflagellates." Journal of Phycology 42: 746-748.
van Oppen, M., Palstra, F., Piquet, A., and Miller, D. 2001. Patterns of coral-dinoflagellate associations in Acropora: significance of local availability and physiology of Symbiodinium strains and host-symbiont selectivity. Proceedings of the Royal Society of London Biological Sciences 268: 1759-1767.
[5] Vidal-Dupiol, J., Adjeroud, M., Roger, E., Foure, L., Duval, D., Mone, Y., Ferrier-Pages, C., Tambutte, E., Tambutte, S., Zoccola, D., Allemand, D., and Mitta, G. 2009. Coral bleaching under thermal stress: putative involvement of host/symbiont recognition mechanisms. BMC Physiology 9: 14.
[6] Warner, M., Fitt, W., and Schmidt, G. 1999. Damage to photosystem II in symbiotic dinoflagellates: a determinant of coral bleaching. Proceedings of the National Academy of Science USA 96: 8007-8012.
Weis, V., Reynolds, W., deBoer, M., and Krupp, D. 2001. Host-symbiont specificity during onset of symbiosis between the dinoflagellates Symbiodinium spp. and planula larvae of the scleractinian coral Fungia scutaria. Coral Reefs 20: 301-308.
Wikipedia.com
Edited by Nicole Hebert, student of Grace Lim-Fong

