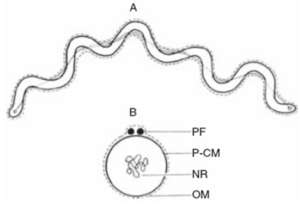
Spirochaeta
Numbered ticks are 11 µM apart.
Gram-stained.
Photograph by Bob Blaylock.
Classification
Domain Bacteria;
Phylum Spirochetes;
Class Spirochetes;
Order Spirochaetales;
Family Spirochaetaceae
[Others may be used. Use NCBI link to find]
Species
|
NCBI: Taxonomy |
Genus species
- Spirochaeta africana
- Spirochaeta alkalica
- Spirochaeta americana
- Spirochaeta asiatica
- Spirochaeta aurantia
- Spirochaeta bajacaliforniensis
- Spirochaeta caldaria
- Spirochaeta coccoides
- Spirochaeta halophila
- Spirochaeta isovalerica
- Spirochaeta litoralis
- Spirochaeta smaragdinae
- Spirochaeta stenostrepta
- Spirochaeta taiwanensis
- Spirochaeta thermophila
- Spirochaeta xylanolyticus
- Spirochaeta zuelzerae
Description and Significance
The genus Spirochaeta represents a group of free-living, saccharolytic non-pathogenic, obligate or facultative anaerobic helical shaped bacteria. Isolated strains have been obtained from a variety of freshwaters and marine waters (Table 1). Extremophilic species include anaerobic, thermophilic isolated form hot springs in New Zealand, the moderately thermophilic Spirochaeta caldaria from cyanobacterial mats of hot springs in Utah and Oregon and the extremely thermophilic Spirochaeta thermophila from marine areas in Shiaskotan Island in Russia. There are also alkaliphilic spirochetes such as S. alkalica, S. africana, S. asiatica isolated from the alkaline Lake Magadi in Kenya and from sulfide-saturated mud sediments of Lake Khatyn in Siberia (Hoover et al, 2003).
In addition, culture-independent studies revealed the presence of Spirochaeta species in other environments such as anaerobic bioreactors, and the digestive tract of termites (Drone et al, 2003). A number of members of the genus Spirochaeta inhabit extreme environments with respect to temperature, salinity, and pressure . These microorganisms may hence harbor enzymes with potential biotechnological applications (Leschine et al, 2006).
Genome Structure
At present, a fully closed genome sequence of a member of Spirochaeta genus is not available. However, the genome size of three species of Spirochaeta are known.
1) Spirochaeta asiatica: 2.1 Mb
2) Spirochaeta alkalica: 2.7 Mb
3) Spirochaeta africana: 2.5 Mb (intro with description)
Using HPLC(High Performance Liquid Chromatography), it was found that the DNA of Spirochaeta species have G+C contents ranging from 51-65 mol% (Wang et al).
Cell Structure, Metabolism and Life Cycle
The morphology and cellular structure of Spirochaeta spp. (and most other Spirochetes) is unique among prokaryotes. The cells are helical in shape and consist of an outer membrane, axial filaments (ultrastructurally similar to bacterial flagella), and a protoplasmic cylinder. The outer membrane or the outer sheath surrounds all of the structures including the axial filaments and the protoplasmic cylinder. The protoplasmic cylinder is the cell body of the organism. It is coiled and is composed of the cytoplasm, the nuclear region and the peptidoglycan-cytoplasmic membrane complex. The axial filaments are usually called the periplasmic flagella but are also known as periplasmic fibrils, axial fibrils and endoflagella. The cell normally has two periplasmic flagella with each one running most of the length of the cell so there is region where the two flagella overlap and gives an arrangement of “1-2-1” meaning that looking at the cell from right to left, there is one flagella at first then two flagella at the region where they overlap and then one again when only the second flagella is present. Points where these flagella attach can be found at the ends of the cell. Figure 2A shows an illustration of a Spirocheata cell showing the outer sheath, the protoplasmic cylinder and the periplasmic flagella. The periplasmic fibrils are found in between the outer membrane and the protoplasmic cylinder (Figure 2B). As mentioned earlier, all species of Spirochaeta have two periplasmic flagella with only one exception, Spirochaeta plicatilis. This species is a large member of Spirochaeta and has 18 to 20 periplasmic flagella inserted near each end of the protoplasmic cylinder. Also, the periplasmic flagella of the spirochete is permanently wound around the cell body and is endocellular. This gives these bacteria a very unique system for motility unlike other flagellated bacteria where their flagella are extracellular and are in direct contact with the environment.The axial filaments reside under the outer membrane and are responsible for the unique mobility behavior of Spirochetes, a trait which has been linked to the pathogenicity of certain genera (Leschine et al, 2006).
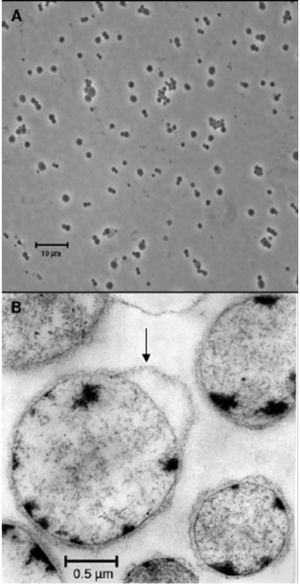
Although the helical shape is typical for members of the Spirochetes phylum, a coccoid shaped Spirochaeta spp. was recently isolated from a termite gut by Droge et al. (2006). A picture of this organism can be seen in Figure 3.
The Spirochaeta genus contains a group of saccharolytic bacteria, responsible for decomposition of (poly)carbohydrates in many aquatic environments. All species of the Spirochaeta genus ferment carbohydrates with formation of acetate, ethanol, CO2, and H2 as major end products. In addition, Spirochaeta isovalerica ferments a number of amino-acids in addition to carbohydrates. All isolated Spirochaeta species utilize the Embden-Meyerhof pathway for glucose catabolization with formation of pyruvate.
Ecology and Applications
Species of the Spirochaeta genus are common inhabitants of a variety of aquatic environments, such as the water, sediments and muds of ponds, marshes, lakes, rivers and oceans. In these environments, Spirochaeta are capable of competing with other microorganisms for available nutrients by exhibiting various types of mobility behaviors. The unique mobility of Spirochetes is due to the helical shape of the cells and the polar positioning of the axial filaments. They are also able to move on surfaces similar to gliding bacteria. And they could move through environments of high viscosities, high enough that most flagellated bacteria cannot.
They are saccharolytic and so can only use carbohydrates as their substrates. They are found in areas where there is high activity of plant decomposition. The sugars, produced as the by-products of the plant decomposition by other organisms, are used by these species through fermentation. The anaerobic species of Spirochaeta, both facultative and obligate anaerobes, grow abundantly whenever cellobiose is available to them. Cellobiose is a product when cellulose is degraded by other organisms. Free living species such as S. aurantia, are able to show strong chemotactic reponse to low cellobiose concentrations and they also have systems that enable them to enhance their chemotactic repsonses to low substrate concentrations in the environment. This gives them an advantage against other cellobiose-utilizing organisms. Another advantage of some species is that they developed strategies to survive in environments with very low concentrations or none at all. Spirochaeta isovalerica does not utilize amino acids as substrates for fermentation. Instead, it catabolizes small amounts of L-leucine, L-isoleucine and L-valine thus forming isovalerate, 2-methylbutyrate and isobutyrate. This strategy also produces ATP which is used as the bacteria's energy source. Other strategies employed by these Spirochetes are metabolism of endogenous RNA or intracellular polyglucose storage granules to produce ATP.
There are symbiotic relationships that were observed in the species, Spirochaeta plicatilis has been associated with Beggiatoa. The spirochete has been found lurking among the trichomes of Beggiatoa. They were found to attracted towards the trichomes through chemotaxis of the metabolites produced by the Beggiatoa which are the substrates of the spirochete. It has also been known that one of its species, Spirocheata caldaria, probably forms symbiotic relationships with the bacteria, Clostridium thermocellum. Spirochaeta caldaria is a thermophilic spirochete taken from a freshwater hot spring. A study conducted by Pohlschroeder et al. showed that cellulose degradation is enhanced in Clostridium thermocellum when the spirochete is present within its environment.
Experiements have shown that some species of Spirochaeta could possibly be used in the direct bioconversion of cellulose-containing wastes to fuel such as ethanol or hydrogen gas (Leschine et al, 2006). Another application of it is that since some species are extremophilic and can survive in high temperatures and high salt concentrations, some of its enzymes can be used.
References
Droge, S., Frohlich, J., Radek, R., Konig, H. "Spirochaeta coccoides sp. nov., a Novel Coccoid Spirochete from the Hindgut of the Termite, Neotermes castaneus". 2006. Applied and Environmental Microbiology. Volume 72. Number 1. p. 392-397.
Hoover, R., Pikuta, E., Bej, A., Marsic, D., Whitman, W., Tang, J., Krader, P. "Spirochaeta americana sp. nov., a new haloalkaliphilic, obligately anaerobic spirochaete isolated from soda Mono Lake in California". 2003. International Journal of Systematic and Evolutionary Microbiology. Volume 53. p. 815-821.
Leschine, S., Paster, B., Canale-Parola, E. "Free-living saccharolytic spirochetes: The genus Spirochaeta". Prokaryotes. 2006. Volume 7. p. 195-210.
Wang, G., Schwartz, I. "Spirochaetes". 2006. Encyclopedia of Life Sciences. John Wiley & Sons, Ltd. p. 1-7.
Author
Page authored by Dieter Maurice Tourlousse and Godfrey Josef Torres, students of Prof. Jay Lennon at Michigan State University.