The role of Bifidobacterium longum in a healthy human gut community
Classification
Kingdom: Bacteria
Division: Actinobacteria
Class: Actinobacteria
Order: Bifidobacteriales
Family: Bifidobacteriaceae
Genus: Bifidobacterium
Species: longum
By Luke Calcei
An Overview of Digestive Health
A healthy human gut is imperative to living a healthy life. Faulty digestion can limit the amount of nutrients extracted from healthy food sources. Unhealthy immune systems lend themselves to having a ‘leaky gut’ [1]. Weak intestinal epithelial cells (IECs) are the cause of leaky guts. Although the intestines are deep within humans, the intestinal barrier is a primary barrier from the external environment. While, it is not typically viewed this way, a leaky gut is similar to having an open wound. Intestinal wounds occur when pathogenic microflora out compete healthy gut microflora. When flourishing, pathogenic microflora release toxins and inflammatory factors, which compromises IECs [1]. Compromised IECs allow those toxins and undigested food particles into the bloodstream, activating the immune system. If this is a continuous problem, the immune system can become overactive causing various autoimmune diseases where the immune system attacks its own cells. Autoimmune diseases include lupus, alopecia, and arthritis and affect approximately 50 million Americans, nearly 20% of the population [2]. Autoimmune diseases are not life threatening, yet they dramatically inconvenience life and distract the immune system, thus making those with autoimmune diseases more susceptible to contracting and being effected by minor illnesses. Autoimmune responses can be triggered by environmental toxins, foreign bacteria, and viruses in individuals who are genetically predisposed [2]. Improving digestive health can decrease autoimmune incidences improving the overall health of populations in developed countries.
Digestive health can be influenced by the foods you eat, which is directly linked to the microorganisms inhabiting the human gut. Bifidobacterium is a genera of bacterium that has been linked to improving digestive health.
The Discovery and History of Bifidobacterium longum
The Pasteur Institute has played a large role in the discovery and knowledge of Bifidobacterium. Bifidobacterium were first discover in 1899 by a French pediatrician, Henry Tissier, who observed a peculiar “Y” shaped microorganism in the stool of infants having diarrhea [3]. Tissier named these microorganisms using the Latin root “bifid” meaning divided by a deep cleft, like the letter “Y” (Figure 1)[4]. Later, in 1907, Nobel prize winning immunologist, Elie Metchnikoff, suggested implanting beneficial bacteria orally would help the digestive system. Tissier and Metchnikoff were the first to introduce the idea of probiotics, ingesting healthy gut microbes to improve overall digestive health.
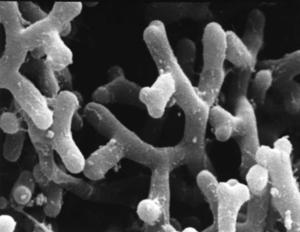
In 2002, three previously separate species of Bifidobacterium merged into one species due to DNA similarities [5]. B. longum, B. infantis, and B. suis became B. longum as the three shared 97% DNA similarities [6]. B. longum subspecies infantis strain 35624 has become the main microorganism related to beneficiary gut function in humans.
An Overview of Bifidobacterium longum
With their interesting “Y” shape, Bifidobacterium longum are considered to be bacilli, rod-shaped, organisms (Figure 1). The use of their unique “Y” is unknown, although it does increase their surface area to volume ratio and could also assist in compartmentalizing cellular structures. B. longum are naturally found in the human digestive system and the vagina [7]. Both of these habitats are hypoxic, which makes sense because B. longum are anaerobic microorganisms. B. longum also lack the catalase enzyme, an enzyme commonly found in aerobic organisms, furthering its anaerobic evidence [7]. B. longum stain gram-positive meaning they have a single cell membrane and a thick peptidoglycan cell wall. The size of B. longum ranges from 4-8µm.
Bifidobacterium longum is more common in the gastrointestinal tract of infants as opposed to adults. Nine out of every 10 bacteria in an infant’s stool sample are B. longum, while only 3% of adult’s gut microbiota is B. longum [8]. This can be explained by the special metabolization B. longum possesses for oligosaccharides. B. longum have a large quantity of genes responsible for transcribing proteins that catabolize a variety of oligosaccharides [7]. Infants' only food source is their mother’s breast milk. There are many oligosaccharides that are unique to and highly concentrated in breast milk [9]. It is logical that B. longum assists infants in harvesting as many nutrients as possible from their lone food source in order to grow and develop normally. Oligosaccharides are short carbohydrate polymers containing 3-10 simple sugars linked through oxygen or nitrogen linkages. Sources of oligosaccharides include breast milk and plant fibers [10]. Since B. longum is located in the vagina, infants are able to acquire this microorganism by passing through the birth canal. As humans mature and their diets diversify, they require a greater variety of microorganisms to assist in digestion. However, in vitro experiments have shown that B. longum can assist IECs to ultimately prevent having a leaky gut [11]. In vivo experiments have also been done with B. longum to show its beneficial effect on the gastrointestinal system [12].
The Role of Bifidobacterium longum in the Human Gut
In order to reap the beneficiary effects of B. longum subspecies infantis strain 35624, one must first get the bacterium into the gut. Since B. longum plays such a large role in digesting oligosaccharides from breast milk, it is logical that humans are born with B. longum in their gut [7][9]. Infants could possibly receive the bacterium through the umbilical cord. B. longum is also naturally found in the vagina, suggesting that the transfer of the bacterium from mother to infant could be ensured through passage through the birth canal. Breastfeeding is another mechanism by which a mother’s gut microbiota is transferred to her child [13]. Babies born via cesarean delivery often lack in breastfeeding while the mother takes antibiotics until her incision heals. These infants miss out on two key factors, which develop their gut microbiota. The early stages of an infant’s life are where the immune system becomes accustomed to foreign bacteria. Infants born via C-section who also lack in breastfeeding are more prone to developing autoimmune diseases, such as asthma, because their immune system does not recognize beneficial gut bacteria when it is later acquired from food [13]. The immune system then becomes hypersensitive, attacking bacteria with the potential to be beneficial. These individuals now suffer from autoimmune diseases due to poor digestive health, which can also cause discomfort. Individuals with a genetic history of autoimmune diseases born via cesarean delivery and lack in breastfeeding are even more likely to suffer severe chronic autoimmune diseases.
However, individuals without a predisposition to autoimmune diseases may be able to develop a healthy gut microbiota through their diet even if they were born via C-section and lack breastfeeding. It is also important for individuals born vaginally who were properly breastfed to maintain a healthy, diverse diet to replenish gut microbial colonies. Since the gut is a hypoxic environment, many foods with beneficial gut bacteria are fermented. Bifidobacterium can be found plentifully in many food types including: goat dairy products, like yogurt, kefir, seaweed, and miso soup [14]. Probiotics, pills filled with millions, sometimes billions, of healthy gut microbes are another mode of acquiring healthy gut microbes, such as B. longum, in to the gut microflora. Recently, the market for probiotics has expanded substantially. In the last five years, per capita spending on probiotic supplements has doubled in the United States and has also grown in Italy, Russia, and Brazil [15]. Similar growth has also been observed in probiotic rich foods, such as yogurt. The issue with acquiring healthy gut bacteria from probiotics versus from food sources is that the gut is an anaerobic environment and therefore many gut microbes are poisoned by oxygen. As little as 10-minute exposure to oxygen can kill some anaerobic bacteria [16]. Therefore, obligate anaerobes and microaerobic organisms could potentially be dead in probiotic supplements, which would not allow the user to reap their health benefits. Acquiring healthy gut microbes is most effective through diet because microorganisms are ensured to be living and are also more likely to enter an exponential growth phase since they have plentiful nutrients from the food source. Knowing how Bifidobacterium longum inhabit and flourish in the human gut is the first step to utilizing microorganisms in a mutualistic relationship.
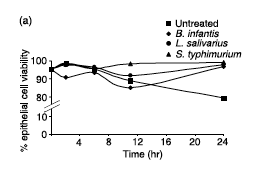
O’Hara et al. performed an in vitro experiment involving intestinal epithelial cells (IECs) and dendritic cells (DCs) exposed to Bifidobacterium longum subspecies infantis strain 35624. In this experiment, human cells were treated with B. longum and other helpful gut bacteria. The control consisted of untreated IECs and DCs, cells without exposure to the probiotics. Inflammatory signals and anti-inflammatory signals from the IECs and DCs were measured using various assay techniques [11]. The researchers measured IEC viability to show a general beneficiary aspect of B. longum on intestinal cells (Figure 2). Twenty-four hours after being treated with the probiotics, IEC viability increased from 80% in untreated cells to nearly 100% in the IECs treated with B. longum [11]. More specifically, flagellin–induced interleukin-8 (IL-8) secretion was measured. IL-8 is a small signaling protein in humans often released by epithelial cells. IL-8 is a primary signaling protein that causes a cascade of effects. One of the cellular responses to IL-8 is the release of histamine, which triggers inflammation [17]. O’Hara et al. showed that exposure to B. longum caused a decrease in IL-8 secretion implying less inflammation in IECs. Interleukin 10 (IL-10) was also measured in DCs in the experiment done by O’Hara et al. IL-10 has an opposite effect on cells compared to IL-8. IL-10 is another small signaling protein, like IL-8, yet IL-10 results in anti-inflammatory responses [18]. Cells treated with B. longum increase IL-10 secretion compared to untreated cells, suggesting less inflammation [11]. The results of this experiment are clear; B. longum has a beneficial relationship with IECs and DCs, although the exact mechanism is unknown. B. longum increases IEC viability, decreases inflammatory signals, and increases anti-inflammatory signals, all traits that promote gastrointestinal health. By preventing inflammation, B. longum prevents the gut from being permeable to pathogenic toxins and food particles. As stated previously, this will prevent the immune system from becoming unnecessarily overactive lessening autoimmune diseases and irritable bowel syndrome [1]. Decreasing the amount of autoimmune occurrences will promote a stronger immune system in general.
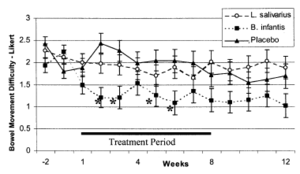
Another experiment was done in vivo by O’Mahony et al. in 2005. This experiment aimed to compare humans with irritable bowel syndrome (IBS) symptoms to their cytokine profiles. In this experiment, 77 subjects previously diagnosed with IBS were given a milk beverage for 8 weeks. Half of the subjects received a probiotic beverage, infused with either Lactobacillus salivarius or B. longum subspecies infantis strain 35624, the other half was provided with a placebo. Individuals recorded their IBS symptoms on a daily schedule. At the beginning and termination of the 8-week period, blood samples were taken and interleukin 10 (IL-10) and interleukin 12 (IL-12) concentrations were measured. Once again, IL-10 is a cytokine responsible for anti-inflammatory responses in humans. IL-12 is a stimulatory protein associated with the immune system. T cells, responsible for immune function in humans, are stimulated to grow and differentiate by IL-12 [19]. Thus high concentrations of IL-12 implicate an overactive immune system. O’Mahony et al. took the ratio of IL-10:IL-12 to measure inflammatory response in the gastrointestinal system. A greater ratio, more IL-10 compared to IL-12, suggests less gut inflammation and therefore a healthier digestive system. A small ratio, more IL-12 compared to IL-10, suggests greater inflammation in the intestines, which makes the gut more permeable to pathogenic toxins and food particles. Pathogens who take advantage of a compromised defense barrier, like a permeable gut, are considered opportunistic pathogens. This ultimately results in an overactive immune system, increasing the likeliness to develop autoimmune diseases. When an autoimmune disease occurs on top of a having a susceptible gut membrane, opportunistic pathogens are more likely to thrive and outcompete healthy bacteria, such as B. longum. Clostridium difficile is a known opportunistic gut pathogen, which commonly outcompetes healthy gut microbiota when antibiotics are administered to the host when suffering from a non-related bacterial infection. The results of this experiment done by O’Mahony et al are quite convincing. B. longum was much more beneficiary to digestive health than the other gut microbe tested, L. salivarius, in terms of both digestive discomfort, as well as physiological cytokine profiles. IBS symptoms that were recorded daily include: bowel movement difficulty, bloating, and abdominal discomfort, all of which improved substantially in subjects provided B. longum over the 8 week test period (Figure 3). Those who received the placebo or L. salivarius also showed improvement, but to a lesser extent than subjects who were given B. longum [12]. The cytokine profiles exhibited similar results. The IL-10:IL-12 ratio increased three fold in subjects provided with B. longum, while the placebo group only showed slight increase [12]. This suggests that B. longum decreased gastrointestinal inflammation in subjects with IBS to similar cytokine levels of healthy individuals. The IL-10:IL-12 ratio in the other treatment group, given L. salivarius, decreased; yet the difference was no greater than the margin of error [12]. All of the results combine to implicate a higher quality of life and less gastrointestinal inflammation in humans with a gut microbiota that includes B. longum. B. longum administration resulted in returning abnormal cytokine levels in subjects with IBS to cytokine levels observed in healthy individuals.This study suggests that diets and probiotics rich in B. longum have the potential to improve the lives of the 50 million Americans suffering from the daily discomfort of IBS [20] and possibly benefit the lives of 50 million more Americans suffering from chronic autoimmune diseases [2].
A healthy digestive and immune system are not the only benefits provided by B. longum in humans. Recent studies by the National University of Ireland, University College Cork (UCC), have shown a correlation between B. longum and the brain. This is not the first connection found between the gut microbiota and the brain. Since 2005, researchers have studied the gut-brain axis, that is, the relationship between the gastrointestinal tract and the nervous system. The gut-brain axis was first discovered in the 1960s when peptides were found in both the gut and the brain [21]. The Head of the Psychiatry department at UCC, Ted Dinan, led the experiment to test the relationship between B. longum, stress levels, and memory. Over a 2-month period, 22 healthy men blindly took either a probiotic containing a billion B. longum or a placebo for a month and then received the other for the following month [22]. Cortisol, a stress hormone, was measured at the beginning of the experiment and at the end of each month. Subjects also completed simple memory tests at these times [22]. During the month of receiving B. longum, subjects displayed improved memory as well as lower anxiety. Suggesting gut microbes can benefit the human brain is a powerful, novel idea, yet many mysteries remain pertaining to the mechanisms between B. longum role in the gastrointestinal tract and the brain.
Conclusion
Humans are hardly human. According to the Smithsonian Magazine, humans carry roughly 37.2 trillion of their own cells [23], while carrying 100 trillion microbes [24]. In terms of mass and volume, nonhuman particles dominate inside the human body. Humans are currently thought to be 90% bacterial and only 10% human [24]. Ever since the discovery of microbial life, by Robert Hooke and Antoni van Leeuwenhoek in 1665, microorganisms have been feared [25]. Furthermore, research done by French Scientist Louis Pasteur in the 19th century proved these microorganisms cause disease, sparking a field of medicine dedicated to holding these microorganisms at bay. Over the last century, soaps, hand sanitizer, antibiotics, and vaccines have all been manufactured to kill microorganisms and exclude them from human life. Yet, within the last decade or so, mindsets have altered among the medical world: some microorganisms are good for us. Humans went from killing all forms of microbial life through nonspecific means to intentionally consuming bacteria in pill form via probiotics. From viruses to bacteria, microbes help humans develop a strong immune system and digest an array of food sources. Some microbes may even improve the human psychological state [22]. Microbes play a large role in benefitting the human life and Bifidobacterium longum is a crucial player among those microbes.
B. longum assists humans in many ways. The presence of B. longum in the gastrointestinal tract of humans has shown to decrease proinflammatory signals, such as IL-8, and increase anti-inflammatory signals, such as IL-10 [11][12]. By regulating intestinal inflammation, B. longum reduces the permeability of the epithelial cells in the gut preventing food particles and toxins from entering the blood stream on a regular basis and causing an overactive immune system. B. longum strengthens the primary barrier of the gut wall from the outside world, making individuals healthier and more resistant to diseases. By lessening the activity of the immune system, B longum has the indirect potential to improve the lives of the millions of Americans suffering from autoimmune diseases by decreasing the overall activity of the immune system. A less active immune system may not cure autoimmune diseases, but it will decrease the rate at which immune cells attack their own cells, thus lessening the severity of autoimmune diseases. B. longum may also improve short term memory and decrease stress levels [22].
Although the exact mechanism of how B. longum produces so many beneficial traits in humans is unknown, there is no denying it acts as a major aid to a healthy human life. The most positive upside of probiotics is that they are harmless and can only result in positive side effects. As knowledge deepens surrounding beneficial bacteria, it will be important to educate the public about how microorganisms can improve healthy living and how individuals can maximize the benefits produced by these microbes through a healthy and diverse diet. With all of the recent breakthroughs in how microorganisms can improve human life, funding will currently be at a premium, so it will be interesting to see how research develops to uncover the precise mechanisms between foreign microbial cells and human cells. Once the various mechanisms are uncovered, humans may be able to utilize this knowledge to further create new and safe ways to mimic these mechanisms for similar results; so stay tuned, microbial breakthroughs have been extraordinary in recent decades.
References
1. Kathy, M. "The Surprising Link between Alopecia and the Gut." OptiBac Probiotics. Wren Laboratories, 9 Oct. 2015. Web. 11 Apr. 2016. <http://www.optibacprobiotics.co.uk/blog/2015/10/the-surprising-link-between-alopecia-and-the-gut>.
2. American Autoimmune Related Diseases Association. "Questions and Answers." AARDA. AARDA, 12 Dec. 2015. Web. 18 Apr. 2016. <http://www.aarda.org/autoimmune-information/questions-and-answers/>.
3. Oyetayo, V.O. and Oyetayo, F. L. 2004. Potential of probiotics as biotherapeutic agents targeting the innate immune system. African Journal of Biotechnology 4:123-127. https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&cad=rja&uact=8&ved=0ahUKEwjyr5GanbDMAhUlroMKHQbAChkQFggfMAA&url=http%3A%2F%2Fwww.academicjournals.org%2Farticle%2Farticle1379957698_Oyetayo%2520and%2520Oyetayo.pdf&usg=AFQjCNGyDMebAZivghDU34h_isLC_XmQLA&bvm=bv.120853415,d.amc
4. "bifid". Dictionary.com Unabridged. Random House, Inc. 20 Apr. 2016. <Dictionary.com http://www.dictionary.com/browse/bifid>.
5. Sakata, S., Kitahara, M., Sakamoto, M., Hayashi, H., Fukuyama, M. & Benno, Y. 2002. Unification of Bifidobacterium infantis and Bifidobacterium suis as Bifidobacterium longum. Int J Syst Evol Microbiol 52: 1945–1951. http://www.ncbi.nlm.nih.gov/pubmed/12508852
6. Mattarelli, P., C. Bonaparte, B. Pot, B. Biavati. 2008. Proposal to reclassify the three biotypes of Bifidobacterium longum as three subspecies. Int J Syst Evol Microbiol 58: 767-772. http://www.ncbi.nlm.nih.gov/pubmed/18398167
7. Schell, M. A., M. Karmirantzou, B. Snell, D. Vilanova, B. Berger, G. Pessi, M. C. Zwahlen, F. Desiere, P. Bork, M. Delley, R. D. Pridmore, F. Arigoni. 2002. The genome sequence of Bifidobacterium longum reflects its adaptation to the human gastrointestinal tract. PNAS 99: 14422-14427. http://www.pnas.org/content/99/22/14422.full
8. Garrido, D., S.R. Moyano, R. J. Espinoza, H.J. Eom, D.E. Block, D.A. Mills. 2013. Utilization of galatoogligosaccharides by Bididobacterium longum subsp. infantis isolates. Food Microbiol 33(2): 262-270. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3593662/
9. Bode, L. 2012. Human milk oligosaccharides: every baby needs a sugar mama. Glycobiology 22: 1147-1162. http://www.ncbi.nlm.nih.gov/pubmed/22513036
10. National Library of Medicine. "Oligosaccharides." U.S National Library of Medicine. U.S. National Library of Medicine, 2011. Web. 25 Apr. 2016. <https://www.nlm.nih.gov/cgi/mesh/2011/MB_cgi?mode=&term=Oligosaccharides>.
11. O'Hara, A., O'Regan, P., Fanning, A., O'Mahony, C., MacSharry, J., Lyons, A., Bienenstock, J., O'Mahony, L., Shanahan, F. "Functional modulation of human intestinal epithelial cell responses by Bifidobacterium infantis and Lactobacillus salivarius." 2006. Immunology 118:202-215.http://onlinelibrary.wiley.com/doi/10.1111/j.1365-2567.2006.02358.x/full
12. O’Mahony, L., J. McCarthy, P. Kelly, G. Hurley, F. Luo, K. Chen, G. C. O’Sullivan, B. Kiely, J. K. Collins, F. Shanahan, E. M. M. Quigley. 2005. Lactobacillus and Bifidobacterium in irritable bowel syndrome. Gastroenterology 128: 541-551. http://www.sciencedirect.com/science/article/pii/S0016508504021559
13. Taylor, Paul. "C-section Babies Missing Crucial Gut Bacteria." The Globe and Mail. Health Navigator, 11 Feb. 2013. Web. 25 Apr. 2016. <http://www.theglobeandmail.com/life/health-and-fitness/health-navigator/c-section-babies-missing-crucial-gut-bacteria-study-finds/article8440728/>.
14. Group, Edward. "Probiotic Foods." Global Healing Center. N.p., 21 Jan. 2011. Web. 25 Apr. 2016. <http://www.globalhealingcenter.com/natural-health/probiotic-foods/>.
15. Watson, Elaine. "US per Capita Spending on Probiotic Supplements Expected to Nearly Double by 2016." Nutra. William Reed Business Media, 2 Feb. 2012. Web. 18 Apr. 2016. <http://www.nutraingredients-usa.com/Markets/US-per-capita-spending-on-probiotic-supplements-expected-to-nearly-double-by-2016>.
16. Jones, Linda B. "Anaerobic Bacteria Culture." Anaerobic Bacteria Culture. National Institues of Health, 5 Apr. 2003. Web. 15 Apr. 2016. <http://www.surgeryencyclopedia.com/A-Ce/Anaerobic-Bacteria-Culture.html>.
17. Hedges, Jones C., Cherie A. Singer, and William T. Gerthoffer. 2000. Mitogen-Activated Protein Kinases Regulate Cytokine Gene Expression in Human Airway Myocytes. American Journal of Respiratory Cell and Molecular Biology, 23: 86-94.http://www.atsjournals.org/doi/abs/10.1165/ajrcmb.23.1.4014#.Vx7MvD-R_R1
18. Eskdale, J., D. Kube, H. Tesch, G. Gallagher. 1997. Mapping the human IL10 gene and further characterization of the 5’ flanking sequence. Immunogenetics 46: 120-128. http://link.springer.com/article/10.1007%2Fs002510050250
19. Murphy, K. M., A. O’Garra, S. F. Wolf, C. S. Tripp, S. E. Macatonia, C. S. Hsieh. 1993. Development of TH1 CD4+ T cells through IL-12 produced by Listeria-induced macrophages. Science 260: 547-549. http://science.sciencemag.org/content/260/5107/547.long
20. International Foundation for Functional Gastrointestinal Disorders. "Facts About IBS." About IBS. Nternational Foundation for Functional Gastrointestinal Disorders, 21 Apr. 2016. Web. 20 Apr. 2016. <https://www.aboutibs.org/site/what-is-ibs/facts/>.
21. Holzer, P., A. Farzi. 2014. Neuropeptides and the Microbiota-Gut-Brain Axis. Adv Exp Med Biol 817: 195-219. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4359909/
22. Sample, Ian. "Probiotic Bacteria May Aid against Anxiety and Memory Problems." The Guardian. Guardian News and Media, 18 Oct. 2015. Web. 19 Apr. 2016. <https://www.theguardian.com/science/2015/oct/18/probiotic-bacteria-bifidobacterium-longum-1714-anxiety-memory-study>.
23. Eveleth, Rose. "There Are 37.2 Trillion Cells in Your Body." Smithsonian. Smithsonian Institution, 24 Oct. 2013. Web. 17 Apr. 2016. <http://www.smithsonianmag.com/smart-news/there-are-372-trillion-cells-in-your-body-4941473/?no-ist>.
24. Sparks, and Honey. "Humans: 10% Human and 90% Bacterial." Big Think. N.p., 22 May 2013. Web. 20 Apr. 2016. <http://bigthink.com/amped/humans-10-human-and-90-bacterial>.
25. Gest, H. 2004. The discovery of microorganisms by Robert Hooke and Antoni Van Leeuwenhoek, fellows of the Royal Society. Notes Rec R Soc Land 58: 187-201. http://www.ncbi.nlm.nih.gov/pubmed/15209075
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2016, Kenyon College.
