Ulcerative Colitis caused by Bacteria?
Introduction
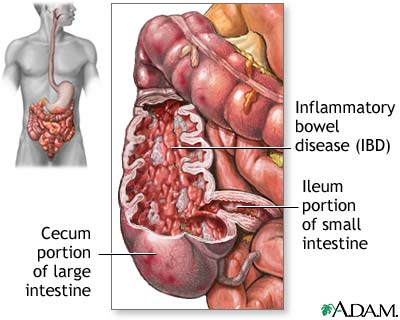
Ulcerative colitis is a complex chronic inflammatory disease that affects the large intestine and the rectum[Figure1]. UC causes inflammation or sores in the lining of the colon and rectum. These ulcers form where inflammation has killed the cells that usually line the colon[13]. Ulcerative colitis can take many forms and depends on the area affected. The four major forms are ulcerative proctitis, proctosigmoiditis, left-sided colitis, and pancolitis[10]. Ulcerative proctitis is the least severe IBD and is defined by inflammation that affects the rectum and does not cover more than six inches. Left-sided colitis is when inflammation of the colon only occurs on the left side. Pancolitis is when there is inflammation throughout the colon. Fulimant colitis is one last form of ulcerative colitis that is life-threatening and extremely dangerous. This type of colitis affects the entire colon, causes severe pain, profuse diarrhea, and can cause dehydration or shock. Serious complications can occur such as toxic megecolon or rupturing of the colon[10]. While the term ulcerative colitis will be used pancolitis will be the form that is focused on because this is the most common form of the disease.
Symptoms of ulcerative colitis include, but are not limited to: abdominal cramps, diarrhea, anemia, weight loss, fatigue, rectal bleeding, loss of appetite, loss of bodily fluids and nutrients, joint pain, as well as others[9]. These symptoms are also common symptoms of other Inflammatory Bowel Diseases (IBD) which makes diagnosis complicated. Also, multiple IBD's can occur at the same time and one can progress into the other further complicating diagnosis.
Ulcerative Colitis is closely related to another IBD, Crohn's disease. Crohn's disease differs from UC in that it affects deeper into the small intestines. Other than that difference the two diseases are very similar. The exact pathogenesis of ulcerative colitis in unknown and remains an important topic of research. Ulcerative colitis most commonly starts in late adolescence to early adulthood, but can occur in young children and elderly people[13]. Ulcerative colitis is found all over the world, but is most common in England, North America, and Northern Europe. Up to 20% of people with UC have a primary relative with the disease or another IBD which suggests that there may be a genetic component to the disease.
Pathogenesis
The exact pathogenesis of ulcerative colitis is unknown. Patients with UC show abnormalities in the response of their immune system. The immune system is chronically activated producing inflammation even in the absence of microbes. Doctors are unsure whether the immune response causes the disease or is in response to the disease. It is likely that no one pathogen or factor is the sole cause of UC. Ulcerative colitis is not caused by specific foods or stress, but these factors may worsen symptoms associated with UC. One common theme researchers find is that patients with UC show elevated levels of microbes in their body. This has been shown in both animal models as well as clinical trials. Mice with targeted deletion of the interlukin-2 gene spontaneously develop Ulcerative colitis in normal conditions, but fail to exhibit the disease in germ free environments[6].
Colonic mucus samples taken from a 12-year-old girl suffering from UC were isolated and studied using cloning and sequencing of the 16S rRNA genes. The sequences were studied using BLAST and the bacteria were assigned to one of the four major phyla:Firmucutes
Bacteroidetes,Actinobacteria,and Proteobacteria. There was a significant difference in the dominant bacteria group of colonic microbiota between the patient with UC and healthy individuals. Several studies show the colonic microbiota of healthy individuals are dominated by Bacteroidetes and Clostridium. The most dominant groups in the study were Proteobacteria and Clostridium. There was also high incidents of Enterobacteriacea and Bacteroidetes fragilis in the UC patient. Also, there was a substantial presence of Pseudomonas aeruginosa in the patient diagnosed with ulcerative colitis. The presence of P. aeruginosa in the feces of a healthy individual has never been reported before. The findings of this study support the hypothesis that bacteria play a role in ulcerative colitis[8].
Another study also supports bacterial involvement in UC. Bacteria from inflamed mucosa were isolated and identified. Twenty bacterial species were isolated from both healthy and diseased individuals. Bacteroides distasonis. B. vulgatus, and Fuscobacterium varium were detected in half the patients with UC. The levels of F. varium were significantly different than healthy individuals. F. varium invades the inflamed mucus and lives in the crypts. The results from this study suggest F. varium is associated with ulcerative colitis[5].br>
Both studies showed that there is a difference in the level as well as the makeup of the intestinal microbiota between ulcerative colitis patients and healthy individuals. While both studies suggest that bacteria play a role in ulcerative colitis they do not claim that the bacteria are the cause. However, the presence of the bacteria most likely enhances the symptoms of the disease.
Diagnosis
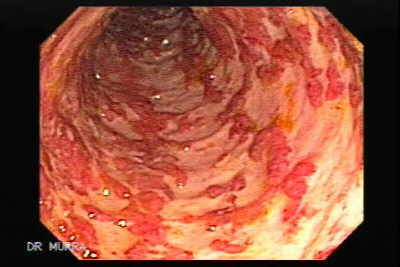
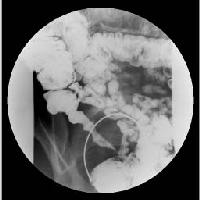
First a complete examination and review of symptoms and medical history of the patient is conducted. If the symptoms suggest an IBD doctors run a series of tests to rule out some IBD's and to see if there is a specific IBD causing the symptoms.
Stool samples are collected to rule out parasites or infection that cause colitis which mimics ulcerative colitis. Blood tests are also run to see if there is anemia, high white blood cell count, or sedimentation. Elevated white blood cell count and sedimentation are indicative of ongoing inflammation in the colon. Stool samples and blood tests can be used to suggest inflammation in the colon, but confirmation of ulcerative colitis requires visualization of the colon.[10] [11]
Colonoscopies allow direct visualization of the colon and can confirm ulcerative colitis. Biopsies of the colonic tissue can be taken during the colonoscopy to determine the severity of the disease[Figure 2].
Another way to confirm the diagnosis of UC is a barium enema X-ray. A chalky substance is inserted in the rectum and injected in the colon. The barium outlines the colon on the x-ray so the colon can be visualized. A Barium enema X-ray is useful, but is less accurate than direct visualization.
CT enterography MR enterography are new imaging methods for visualizing the digestive system[Figure 3]. CT enterography combines a CT scan with contrast to image the small bowel. CT enterography is used specifically for diagnosing Crohn's disease, but is also useful in diagnosing UC. MR enterography shows images of the bowel without using radiation. The tests and procedures that are used to diagnose ulcerative colitis may differ depending on the physician's preference and the equipment and diagnostic center availability [10][11].
Inflammation
The inflammatory response is part if the immune system's response to infection or foreign microbes. The inflammatory response at the cellular level is one of the first responses of the immune system. The first step is the bacteria infecting tissue. Next, macrophages engulf bacteria and release chemical mediators. The chemical mediators cause cytokines to induce selectins on capillary endothelia that bind to neutrophils. The first selectins produced are P-selectin and E-selectin. The selectins grab neutrophils from the bloodstream and slow them down and cause them to roll along the endothelium. These neutrophils are activated to produce and display integrin adhesion moelcules on their surface. Two adhesion molecules intracellular adhesion molecule 1(ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1). Upon binding these molecules stop the neutrophils from moving and initiate extravasation in which the white blood cells squeeze through the endothelial wall and into tissues where the white blood cells can help the macrophages fight off invading microbes. Bradykin is released which loosens junctions to allow extrvasation and triggers prostaglandin synthesis. The final step involves peptides from the bacteria and chemical signals from infected tissues being released and attracting neutrophils to attack the foreign microbes. This inflammation response helps to fight of the infection[7].
Inflammation is an important aspect of the immune system. However, chronic inflammation while trying to repair will cause permanent damage. This is the case with ulcerative colitis. The chronic inflammation is an autoimmune reaction that may be stimulated by intestinal microbiota[13]. During chronic inflammation there is continual recruitment of macrophages and lympohcytes from the circulation. A granuloma forms to try to close of the site of inflammation. Granulomas start as a collection of mononuclear inflammatory cells surronded ny lympohcytes. Fibroconnective tissues deposit around the lesion causing fibrosis or tissue hardening[7]. Granulomas are very common in patients with ulcerative colitis
Since chronic inflammation is the main problem associated with ulcerative colitis this is a key target for treating the disease. Many medicines focus on reducing the inflammation either at the cellular level or by supressing the immune system.
Treatment
There are several different treatment option for patients with ulcerative colitis. The disease is managed from patient to patient and what works for one patient may not work for another patient. Based on the severity of the inflammation a doctor will recommend the treatment or option that is believed to work the best. More often than not there is not just one treatment that a patient will receive. The treatments are adaptive and change with the severity of the inflammation as well as how the inflammation responds to the treatment. The treatment plans are unique to each individual patient and that patient's needs. Sometimes very mild forms of UC can be controlled with lifestyle and diet changes. However, most UC patients require some form of medication. There are several different medications that are prescribed:
Anti-inflammatory drugs are often the first treatment option. These drugs include Sulfazine,Mesalamine, Balsalazide, and corticosteroids. Sulfazine can be effective, but there are many side effects. Mesalamine and Balsalazide are usually prescribed as the first line medications. Both medications work to reduce the inflammation in the colon. The exact mechanism of how the inflammation is reduced is unknown. These medicines have fewer side effects than sulfazine.
Corticosteroids are prescribed to reduce inflammation in the colon, but are not intended for long term use because of the side effects associated with them. Corticosteroids are used when there is an intense flare-up of inflammation to reduce the inflammation to a more manageable level[10].
Immune system suppressors are also used to reduce inflammation, but target the immune sytem, rather than just treating the inflammation [10]. Azathiprine and 6-Mercaptopurine suppress the immune system slightly and reduce bowel inflammation.These medications take a longer time to start working so they are sometimes paired with a corticosteroid. Anoter immunosuppressor is Inflixmab which is prescribed to patients with moderate to severe ulcerative colitis that has not responded to other treatments. This medicine can reduce the need for surgery by almost 50%[10].
Surgery is used as treatment when UC has not responded to other treatments or precancerous changes develop in the colon. Approximately 25-30% of patients with UC end up needing surgery. Ileoanal anastomosis is the most common surgery for UC. The entire colon and the diseased lining of the rectum are removed. The primary advantage of Ileaonal anastomosis is that waste can still be eliminated normally afterward and the surgery can be done laproscopically. Proctocolectomy with ileostomy is also another common surgery for UC patients which also involves removing the colon and the rectum. However, a stoma (opening) is created on the outside of the body and waste is eliminated through the stoma into a bag. Complications of this surgery include infections and complications with the stoma [10].
Prognosis
Ulcerative colitis is a chronic disease consisting of multiple flare-ups followed by periods of remission. The goal of most treatment options is to reduce the flare-ups and prolong the periods of remission. Rapid initial inflammation may result in more severe complications. Very rarely does total remission occur after only one flare-up. If this occurs the inflammation was more likely due to an infection in the colon and not UC. People with ulcerative proctitis have the best prognosis. Individuals with ulcerative colitis that affects the entire colon with more severe inflammation have the worst prognosis. As stated before 25-30% of patients require surgery at some point in their life. The main advantage to surgery is that the surgery results in curing of the disease. Complications of ulcerative colitis include, but are not limited to severe bleeding, perforated colon, severe dehydration, liver disease, osteoporosis, inflammation of skin, an increased risk of colon cancer, and toxic megacolon [10]. Toxic megacolon is rapid widening of the large intestine in one hour to a few days. Most often toxic megacolon results in surgery and removal of the colon. Approximately 5% of patients with ulcerative colitis develop colon cancer. The risk of cancer increases with duration of the disease as well as how severe the damage to the colon is. The risk of cancer in patients with the entre colon affected is up to thirty two times more than healthy individuals [8]. Precancerous changes called dysplasia can occur in the cells lining the colon and colonoscopies every one to two years are suggested for patients with IBD in order to screen for dysplasia.
Genetic component
There seems to be a genetic component that contributes to ulcerative colitis. Twenty percent of patients with ulcerative colitis have a family member of relative with either ulcerative colitis or crohn's disease. Recently multiple studies have used genome wide association scans to investigate genetic susceptibility to ulcerative colitis. Approximately thirty genes have been identified that may increase susceptibility to UC. These genes include immunoglobin receptor gene FCGR2A, 5p15, 2p16, ORMDL3, ECM1, as well as regions on chromosomes 1p36, 12q15, 7q22, 22q13, and IL23R [10].At this point it is unclear whether the identification of these genes will help in treatment with the disease, but may help with determining the pathogenesis.
One research experiment looked at the genetic component of ulcerative colitis and used mice as a model organism. In mice oral administration of dextran sulfate sodium (DSS) results in both acute and chronic ulcerative colitis in mice. Using this model differential susceptibility to DSS was tested in mice. Various inbred strains of mice were used to test susceptibility. After twenty one days of being exposed to DSS the mice were euthanized and samples from the cecum and colon were evaluated. A pathologist examined the samples and scored them for lesions based on severity, ulceration, hyperplasia, and area affected. The study concluded that there were significant differences in susceptibility to DSS among different strains[Figure 3]. The strain differences will be useful for design in the genetic mapping to identify genes that determine susceptibility to DSS-induced colitis. The identification of the genes may predict pathways for intervention in humans. This study further supports the notion that there is a genetic component of ulcerative colitis[4].
<br< Further research is needed to determine the mechanism in which the identified genes result in greater susceptibility to ulcerative colitis. The mechanism may provide a future target for medicine.
Research
There are many potential areas of research for ulcerative colitis. With the pathogenesis being unknown this presents a major area of research. There is a lot of debate whether the pathogenesis is from bacteria, genetics, or both. Another significant area for research is exploring new clinical treatments of the disease in the form of drugs or other potential treatments. Research is also being done on tools for diagnostics such as the recently developed CT enterography.
One recent study looked at whether sigmoidoscopy was sufficient for evaluating inflammatory status of ulcerative colitis patients. The researchers wanted to investigate this question because the area of the most inflammation has not been determined. This suggests that only looking at one area of the colon or inserting in only one area may not cover all the areas of inflammation. The area where the most inflammation is localized may not be covered by just simple sigmoidoscopy. Researchers studied and analyzed medical charts and endoscopic images of UC patients with insertion into the cecum. The severity of inflammation was analyzed using the Mayo score of endoscopic index with 0 being normal or inactive, 1 being mild disease, 2 was moderate disease, and 3 indicates severe disease. Two thirds of the patients had inflammation in the rectum and more than half the patients had negative activity(no inflammation) in descending or proximal portion of the disease. The study did however find that severe inflammatory disease was frequent in the proximal or right side of the colon. The study concluded that since there was a substantial portion of patients with severe inflammation in the right side of the colon sigmoidoscopy was insufficient in evaluating ulcerative colitis in patients and full colonoscopy should be conducted[3].
Another experiment looked at further identifying the microbiota of the gastrointestinal tract that play a role in ulcerative colitis. Researchers performed independent rRNA sequence analysis of gastrointestinal samples from UC and crohn's disease patients as well as healthy individuals who served as a control. Researchers also used phylogenetic analysis to address the possibility that different subpopulations exist within IBD classification. Figure 4 shows the phylogenetic tree analysis from their experiment. The distribution of the microbiota was similar to previous findings in that the microbiota were found in the four major phyla, Bacteroidetes, Firmicutes, Proteobacteria, and Actinobacteria. Ulcerative colitis and crohn's disease patients had more Proteobacteria and the Bacillus group of Firmicutes. The study also found that the microbial populations between the large and small bowel do not differ significantly, but the relative proportions between which phyla are present do differ. Their results were consistent with previous research in that they failed to identify any individual species that were elevated enough to suggest an active etiologic agent. Researchers also found that UC and CD patients manifest in different forms that can be differentiated by microbial populations. This shows a further difference between ulcerative colitis and crohn's disease. This study also found that commensal microbiota decreased in UC and CD patients when compared to healthy individuals. The researchers suggest that there is a staggering level of microbial diversity in the human gastrointestinal tract that remains to be characterized. Further investigation of these microbes may result in finding a microbe or group of microbes that are responsible for the disease[2].
A recent study looks at genetics as the form of pathogenesis. Researchers carried out a genome-wide scan to look for changes in the genetic code common amongst patients with UC. Genetic data was compared between patients with UC and healthy individuals. The researchers found twenty nine new links between a region of the genome and Ulcerative Colitis. Their findings push the total number of genes associated with ulcerative colitis to forty-seven. These genes may play an important role in ulcerative colitis. The researchers also looked at whether these regions overlapped with regions found in a similar study on crohn's disease and found that at least nineteen of the forty-seven overlapped. Many of the overlapping regions included genes involved in maintaining a group of T-cells involved in human immune response. This supports the notion that the way in which the immune system responds to bacteria in the gut is an important part of ulcerative colitis. The significance of the findings of the study is that there are more genetic pathways that can be explored in order to find new treatments for UC. While these findings are supportive there is still much research that must be done in order to treat UC at the genetic level[1].
Conclusion
Ulcerative colitis is a complicated chronic inflammatory condition that affects many people. The range and severity of the disease depends on the patient. The exact pathogenesis of UC is unknown but there seems to be a genetic factor as well as a microbial factor. Research has shown raised levels of Proteobacteria as well as other microbes. However, no single microbe or group of microbes has been identified as the sole cause of ulcerative colitis. Diagnosis of UC includes a review of symptoms, stool samples, and some form of visualizing the colon whether a colonoscopy or the newer forms of CT and MR enterography. Complications of diagnosis occur because of shared symptoms with crohn's disease. Treatment for UC includes taking medications that reduce inflammation and in the most severe cases surgery. The prognosis for ulcerative colitis has improved and continues to improve. The most serious complication of UC is an increased risk of colon cancer. However, diligent monitoring of the disease with colonoscopies can help prevent or detect colon cancer early. Since the pathogenesis is unknown it presents a major area of research for the disease. Research is being conducted on the two major hypotheses of cause of UC: genetic components and microbial involvement. There have been gains in both areas of research, but there is still an abundant amount of room for future work in both areas. There is hope in the recent findings from research that one day ulcerative colitis can be cured without the need of surgery.
References
[1] Anderson, C., Boucher, G., Lees, CW., Franke, A., D'Amato, M. Meta-analysis identifies 29 additional ulcerative colitis risk loci, increasing the number of confirmed assoaciations to 47. Nature Genetics. 2011.
[2] [Frank, D., Amand, A., Feldman, R., Boedeker, E., Harpaz, N., Norman, R. Molecular-phylogenetic chacracterization of microbial community imbalances in human inflammatory bowel disease. National Academy of Sciences of the USA. 2011. Volume 104. Issue 34. p, 13780-13785. ]
[3] [Kato, J., Kuriyama, M., Hiraoka, S., Yamamoto, K. Is sigmoidoscopy sufficient for evaluating inflammatory status of ulcerative colitis patients? Journal of Gastroenterology & Hepatology. 2011. Volume 26. Issue 4. p. 683-687.]
[4] [Mahler, M., Bristol, I., Leiter, E., Workman, A., Birkenmeier, C., Elson, O., Sundberg, J. Differential susceptibility of inbred mouse strains to dextran sulfate sodium-induced colitis. Am J Physiol Gastrointest Liver Physiol. 1998. Volume 274. p. 554-551.]
[5] [Ohkusa, T., Sato, N., Ogihara, T., Morita, K., Ogawa, M., and Okayasu, I. Fusobacterium varium localized in the colonic mucosa of patients with ulcerative colitis stimulates species-specific antibody. Journal of Gastroenterology and Hepatology. 2002. Volume 17. p. 849-853]
[6] [Sellon, R., Tonkonogy, S., Schultz, M.,Dieleman, L. Grenther, W., Balish, E., Rennick, D., and Sartor, R. Resident Enteric Bacteria Are Necessary for Development of Spontaneous Colitis and Immune System Activation in Interleukin-10-Deficient Mice. Infect Immuniology. 1998. Volume 66. p. 5224-5231.]
[7] Slonczewski, Joan L. and Foster, John W. Microbiology:An Evolving Science. Ed 2. W.W. Norton:2011.
[8] [Wang, M., Molin, G., Ahrne, S., Adawl, D., Jeppson, B. High proportions of Proinflammatory Bacteria on the Colonic Mucosa in a Young Patient with Ulcerative Colitis as Revealed by Cloning and Sequencing of 16s rRNA Genes. Dig Dis Sci. 2007. Volume 52. p. 620-627.]
[9] [1]
[10] [2]
[11] [3]
[12] [4]
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2009, Kenyon College.