User:Liwoo92
Introduction
The use of antibiotics has become common in the livestock production around the world. The growth-promotic effects of antibiotics are undisputed, but the collateral and long-term effect are a cause for a heated debate and banning in the European Union in 2005(OMAFRA, 2005.) Antibiotics increase the efficiency of animal growth by inhibiting the growth of microbes in the gastrointestinal tract which triggers immune responses in the host (Gaskins et al., 2002). They have been shown to improve the health of animals raised in close quarters in conventional operations and also reduce microbes on the meat that cause foodborne illnesses (OMAFRA, 2005). However, there is much concern regarding the development of antibiotic resistance associated with the use of drugs. It is important to study the microbial system within the host organism to carry out further studies related to the controversy.
Host Microbial Community
All livestock harbor a intestinal microbes in a dense and highly diverse community, which are engaged in complex interactions with one another. Despite the diversity, specific animals have innate microbial communities. For example, in swine, major groups include Bacteroides, Peptostreptococcus, Bifidobacterium, Selenomonas, Clostridium, Butyrivibrio, and Escherichia (Moore et al., 1987). Most of the microbes are found in the large intestine because of slow digesta turnover. A low number of microbes occupy the small intestine because of low pH and the rapid digesta flow which results in bacterial washout (Gaskins, 2000). Gut flora benefits the host in a variety of ways including digestion of unutilized energy substrates, stimulating cell growth, repressing the growth of harmful microorganisms, training the immune system to respond to pathogens, and defending against some diseases (Guarner et al.,2003). The gut flora and the host form an important mutualistic relationship.
Immunological Interactions
The non-pathogenic microbes benefit their host by stimulating the development of immune responses. The host organism develops defensive responses such as the constant mucus production and high cell turnover of the GI tract (Gaskins et al., 2002). The immune responses result in bacterial washout, controlling the growth rate of the enteric bacteria (Gaskins et al., 2002). The washout leads to the prevention of the pathogen growth, defending against diseases. Also, indigenous bacteria are proposed to prevent the colonization of nonindigenous bacteria via competition for nutrients and mucosal attachment sites, or alteration of the growth environment by producing antimicrobial compounds and modified bile acids (Rolfe, 1997). Studies with germfree animals show that the absence of indigenous bacteria leads to an underdeveloped immune system and less effective response to pathogens (Gaskins, 2000). Thus, the normal intestinal microbes provide the host with important defense by outcompeting pathogenic bacteria and preventing enteric diseases (Nisbet, 1998).
Immunity vs. Growth efficiency
However, those innate immune responses are offered at the expense of the growth efficiency (Gaskins et al., 2002). Building the defense against the microbial community in the GI tract requires disproportionate amounts of energy and resources. For example, in swine, although the GI tissues only represent 5% of the total body weight, they receive 15 to 35% of the whole body oxygen consumption and protein turnover because of high metabolic rate (Gaskins, 2000). Germfree organisms do not have to develop such immune responses, and instead, they can make effective use of their energy by investing it in weight growth.
Nutrients
Microorganisms require nitrogen and phosphorus for decomposition of hydrocarbons and incorporation into biomass, therefore the availability of these nutrients are important for bioremediation. Still, the nutrient availability depends heavily on the physical status of oil spill, whether it is in soil or marine, whether it is in dissolved forms or slick. However, in most studies, there seems to be insufficient nutrients to carry out best degradation performance. Many researchers have reported that the amendment of nutrient can significantly improve the degradation of petroleum pollutants both in water and soil (Xu and Obbard 2004, Liang et al. 2011; Tyagi et al. 2011)
Oxygen
The major degradation pathways for petroleum hydrocarbons involves oxygenates and molecular oxygen, indicating the (Boufadel et al. 2010) importance of oxygen for oil degradation microbes. There are anaerobes capable of degradation, but the rate is significantly lower. For instance, within anoxic basins or sediments, the hypolimnion of stratified lakes, and benthic sediments, oxygen may severely limit biodegradation. Oxygen consumption by oil-degrading bacteria is high, even in some environment such as the water surface where oxygen is readily available right after the oil spill happens, oxygen may rapidly become limiting factor due to slow oxygen diffusion rates and its limited solubility in water. Since most of the degradation is aerobic processes, maintenance of aerobic conditions is important in the bioremediation (Atlas 1991)
Salinity and pressure
The influence of several other environmental factors on hydrocarbon biodegradation has been studied. Typically these factors are associated with the localized environment features such as saline lakes or deep seas where high hydrostatic pressure exists. Studies have shown that generally the biodegradation rate decreases as salinity increases. The deep benthic zone in marine ecosystems has been found to be one of those with least microbial activity, partly due to the higher pressure that limits the microbial activity (Shin and Pardue 2001).
Microbial communities
Microbial community composition
A very diverse group and microbes have been showed to have the ability to degrade petroleum hydrocarbons (Atlas 1981, 180-209), among which bacteria and fungi appear to be the prevalent hydrocarbon degraders in oil contaminated ecosystems.
Bacteria and archaea
Many bacterial strains have been reported to have the ability to degrade recalcitrant compounds in petroleum. More than 20 genera of hydrocarbon degrading bacteria has been isolated including Alpha-, Beta- and Gammaproteobacteria; Gram positives; Flexibacter–Cytophaga–Bacteroides (Teralmoto et al. 2009). On the other hand, fast development of molecular microbiological tool has enabled the identification of many un-culturable microbes and therefore extended the list of microbial species with petroleum hydrocarbon degrading abilities. Species of Pseudomonas, Mycobacterium, Haemophilus, Rhodococcus, Paenibacillus and Ralstonia, are some of the most extensively studied bacteria for their bioremediation capability.
Archaea have been detected in several oil-containing environments, such as petroleum reservoirs, underground crude oil storage cavities, and hydrocarbon-polluted aquifers. Archaea have also been found in oil contaminated environment degrading petroleum hydrocarbons. Lots of Archaea species are found in oil reservoirs, such as Thermococcus celer, Pyrococcus lithotrophicus, Archaeoglobus fulgidus (Ollivier et al. 2000;de Brito et al. 2004).
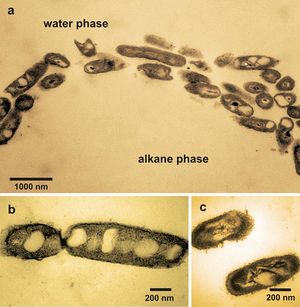
As the molecular biology are developing, scientists and engineers are probing more and more into the mechanisms and genetic traits of these oil-eaters, and are expecting to better utilize them to clean up oil spill faster and more efficient. Pseudomonas species is one of the most common species that have been isolated from oil spill bacteria. The genetic information for hydrocarbon degradation has been found to occur on plasmids (Vila et al. 2010) and this genetic trait shows potential to be transferred to other microbes to make some potential “supermicrobes”. Another interesting bacterium, Alcanivorax borkumensis, as shown in the Figure on the right, is the first carbonnoclastic bacterium that has been sequenced (Santos et al. 2006) in order to get valuable insights into its unusual metabolic capability in oil hydrocarbon degradation.Although barely detectable in unpolluted water body, it blooms right after an oil spill. It was found in oil spills from Alaska (Exxon Valdez) to the Mediterranean waters near Spain (Prestige). It has specific ability to both break down the alkanes that make up part of the oil, and spread a biosurfactant that helps make oil more bioavailable to other microbes. These unique combination of these features provides this microbe with a competitive edge in oil-polluted environment and therefore it was of special interest of many scientists.
Fungi
Fungi, including yeast and filamentous fungi have also been found to have the ability to degrade petroleum carbon. Species reported include Candida, Rhodotorula, Saccharomyces, Sporobolomyces, and Trichosporon etc. (Liu et al. 2009; Atlas 1981).
Algae and cyanobacteria
Interestingly, the ability to oxidize petroleum hydrocarbons is widely distributed, not only among bacteria and fungi, but also among the cyanobacteria and algae. Cerniglia et al. found that the ability to oxidize aromatic hydrocarbons is widely distributed among algae and cyanobacteria. They tested the capability of naphthalene degradation among nine cyanobacteria, five green algae, one red alga, one brown alga, and two diatoms. Results showed that Oscillatoria spp., Microcoleus sp., Anabaena spp., Agmenellum sp., Cylindretheca sp.,Coccochloris sp., Ulva sp., Nostoc sp., Aphanocapsa sp., Chlorella spp., Dunaliella sp., Chlamydomonas sp., Amphora sp., Porphyridium sp., and Petalonia are all capable of oxidizing naphthalene(Gibson et al. 1980). Except various low molecular weight organics, algae also showed ability to degrade high molecular weight PAHs, such as benzo[a]pyrene(Naidu and Juhasz 2000). Beside single compounds, some researchers also investigated the algae/cyanobacteria capability of degrading the whole matrix of oil. For example, Gamila and Ibrahim (Ibrahim and Gamila 2004) valuate the potential role of freshwater isolated algae strains in biodegradation of crude oil (Egyptian light crude oil, specific gravity 0.85) though an algal bioassay. They found that the diatom strain, Nitzschia linearis, and green alga Scenedesmus obliquus treated with 0.1% crude oil showed almost similar capability for degradation of the petroleum hydrocarbons. Interestingly, they also observed the effect of the crude oil on the morphological characters of these two algae. The most pronounced feature is that the algal cells in both strains were aggregated in clusters containing oil drops between their cells, forming an abnormal shape comparing with control culture.
Microbial community dynamics
Both laboratory and field studies have shown that hydrocarbon contamination shifts the overall microbial community structure (Cerniglia et al.1980). Once the site is contaminated, the microbial community composition will be greatly changed both in quantity and composition. Populations of hydrocarbon-degraders normally constitute less than 1% of the total microbial communities, but when oil pollutants are present these hydrocarbon-degrading populations increase, Certain hydrocarbon-degrading taxa become dominant in oil-impacted environments because of natural selection resulting from the pressure of oil contaminants typically to 10% of the community, or even 100%(Tyagi et al. 2011).
Microbial processes
Aerobic processes
Oil is a complex mixture of various components, with different affinity with microbes. The biodegradability of the oil components generally decreases in the following order: n-alkanes, branched-chain alkanes, branched alkenes, low molecular-weight n-alkyl aromatics, monoaromatics, cyclic alkanes, polycyclic aromatic hydrocarbons (PAHs) and asphaltenes (Atlas 1981). No matter what form the component is, these compounds are eventually converted to carbon dioxide and water by microorganisms if in aerobic degradation process, the most common biodegradation. The initial steps in the aerobic biodegradation by bacteria and fungi involve the oxidation of the substrate by oxygenases for which molecular oxygen is required. Aerobic conditions are, therefore, necessary for microbial oxidation of hydrocarbons in various environments. The availability of oxygen in soils, sediments, and aquifers is often limiting and dependent on the type of soil, whether the soil is aerated, weather the water is well mixed etc.
Anaerobic processes
Anaerobic degradation exists, however, the rate of which is very low and the ecological significance appears to be minor. Anaerobic respiration of oil hydrocarbons can be found in the sediments or anoxic zones, where Mn+2, Fe+2 can be the terminal electron acceptor (Atlas 1991; Essaid 1995).
Nitrogen fixation
As previously described, nutrients are important for oil-degrading microbes to survive and therefore fertilization with nitrogen can stimulate the bioremediation of oil contaminated sites. However, in natural environment, there is another process which can bring extra nitrogen to the microbes-nitrogen fixation. Prantera et al. found that two bacterial strains, which belongs to the genera of Agrobacterium and Alcaligenes respectively exhibited ability to degrade gasoline aromatic compounds and fix nitrogen at the same time (Drozdowicz et al. 2002). Chen et al. (1993) examined six species of free-living nitrogen fixing bacteria, Azomonas agilis, Azotobacter chroococcum, Azotobacter vinelandii, and Beijerinckia mobilis for their ability to grow and fix N2 using aromatic compounds as sole carbon and energy source and the result showed that all six species grew and expressed nitrogenase activity on oil components such as benzoate. Except nitrogen fixation by heterotrophic growth, autotrophic growth is also an important process for nitrogen fixation in oil contaminated sites. Phototrophic N2-fixing bacteria such as Pseudomonas stutzeri, Azospirillum brasilense, Aquaspirillum sp.(Eckford 2002) have been identified and isolated from oil contaminated soils, snow, sea ice in Antarctic. Musat et al. (2006) even found that phototrophic cyanobacteria are the main contributors of fixed nitrogen to oil-contaminated and pristine sediments if nitrogen is a limiting factor and if light is available. As a consequence, the oil-degrading heterotrophic community may receive a significant part of combine nitrogen from cyanobacteria.
Current Research
1. A study of the microbial community composition and functional genes in oil contaminated soil
In order to compare microbial functional diversity in different oil contaminated soils, and to find the relationship between the contamination and environmental factors, Liang et al. analyses soil samples from 5 different oil filed and used GeoChip to evaluate the microbial functional genes. Results showed that the samples were clustered by geographic locations and the contaminant degradations genes presented similar patterns under oil contaminant stress. Canonical analysis results also indicated that the local environmental variables significantly affect the microbial functional patterns (Liang et al. 2011).
2. A case study of the environmental condition after Exxon Valdez oil spill
In order to evaluate the long-term environmental effect of Exxon Valdez oil spill in 1989, a series of measurements of the background concentration of nutrients, dissolved oxygen (DO), and salinity were obtained from a contaminated beach. Results showed that both nutrients and DO are limiting factors for biodegradation. Also, the lowest nitrate and DO values were found in the oiled pits, implying that the microbial oil degradation was probably under anoxic conditions associated with denitrification (Boufadel et al. 2010).
3. The application of bioautmentation
The feasibility of a bioaugmentation strategy based on use of microbial formula tailored with selected native strains to remediate diesel contaminated site was assessed. The biodegradation process of diesel oil was assessed by monitoring the DO composition, CO2 evolution rate, microbial load and composition of the community by T-RFLP, physiological profile in Biolog® ECOplates and ecotoxicity. The mixture of microbes that combines 10 bacterial strains selected for resistance to heavy metals was found to efficiently facilated and speed up the bioremediation of diesel hydrocarbons and heavey metals (Alisi et al. 2009)
References
19. Prince R (1993) Petroleum spill bioremediation in marine environments. Crit Rev Microbiol 19:217-242
Edited by Yan Zhou, a student of Angela Kent at the University of Illinois at Urbana-Champaign.