Co-Evolution of Microbes and the Mammalian Gut
By Joanna van Dyk (Kenyon '22)
Introduction
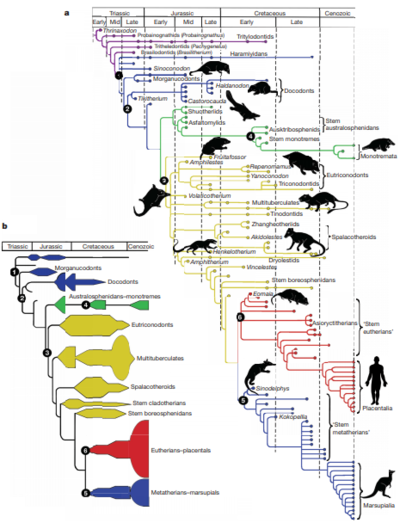
Microbes likely had commensal relationships with the ancestors of mammals, long before they evolved to give birth to live young or obtained many of the traits that characterize the class of vertebrates today[2]. The digestive tract of a mammal provides a perfect environment for microbial colonization, it is moist, warm, and has a continuous supply of food. The microbes, in turn, are able to produce toxin-digesting enzymes that allow their hosts to consume a wide variety of nutrients[3]. In fact, vertebrate guts have provided such a unique and hospitable habitat for microbial colonization, that microbial gut populations are more distinct from "external" habitats (such as lakes and soils) than these habitats are from each other[2]. Today, the microbiota of mammalian guts show similarities between species from similar ancestry, but also between those that have similar diets[2].
The earliest mammals were carnivorous, and without microbes, they would have not been able take advantage of the diverse array of nutrients offered by the plant kingdom. This allowed them to fill many of the ecological niches left when the dinosaurs became extinct 65 million years ago.[4] Mammalian gut microbiomes have evolved to aid their hosts in the digestion of plant toxins that would otherwise completely disable them, such as Nordihydroguaiaretic acid. This chemical would cause desert woodrats (N. lepida) to develop kidney cysts and liver damage if its gut bacteria did not digest it for them. [5] Unlike other vertebrates, mammals are in the unique position of being able to inherit their microbiomes from their mothers through the vaginal canal and through breast milk (see also: Birthing method and infant gut microbiota). This may enable the preservation of microbial lineages over time[6]
This page investigates the evolutionary relationships between mammals and their gut microbiota. It also addresses the questions; how specialized are the microbe lineages associated with mammalian guts? And, when a mammal adopts a different diet, how much does its gut microbiome resemble the microbiomes of its close relatives?
Generation of Evolutionarily Stable Mutualism
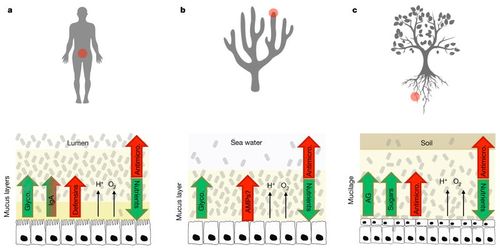
Why do symbiotic bacteria evolve traits that benefit their host?
Although the mammalian gut may provide a warm, hospitable environment for microbes, it is subject to continuous turnover [8][9]. The gut microbiome is full of hundreds of different microbial species that are always in flux, as food passes through the digestive system and non beneficial species threaten to disrupt the community[10]. Because this environment is so competitive and diverse, any mutation that comes at a fitness cost to the microbe is unlikely to survive in the gut even if it is beneficial to the host[7]. Evolutionary theory predicts that natural selection should favor microbes that invest in their own reproduction rather than benefiting the host[8]. There is evidence that hosts have evolved several mechanisms to manipulate the competition pressures between microbes and select for beneficial bacteria in their guts themselves to combat this. Mammals compartmentalise their microbiota with mucus-covered epithelial layers that prevent most organisms from invading the rest of the body[11][12]. They also release antimicrobial peptides into this mucus (antibodies such as immunoglobulin A), which discourage growth when it is too close to the epithelium[11]. The mammal immune system can monitor the species of microbes in the gut through toll-like receptors when the intestine is breached by a pathogen (or harmless microbe)[13], it can also link the beneficial or harmful products of microbial metabolism to the microbes producing them and produce a localized immune response to (potentially) control them[14]. As well as negative selection via localized immune responses. Mammals may also be able to positively select for bacteria by incorporating complex glycans into their epithelial mucus that encourage growth of bacteria able to digest complex carbohydrates, such as Bacteroidaceae[15]. Epithelial mucus can provide an ecological refuge for slow-growing bacteria that may fail to compete with other microbes in the intestine[9].
Colonization and Colonization Resistance of the Gut
Is protection of the host an evolved function of microbial symbionts? Mammals and their microbiota are closely intertwined. Because of this, it is possible that hosts have been able to evolve traits that select for bacteria which protect against pathogens (i.e. make the gut more resistant to pathogen colonization). This makes colonization resistance a combination of both the competitiveness of the established microbiome and the host’s ability to control this microbiome[16]. One study (Buffie et al., 2014) found that as few as one species of bacteria is responsible for protecting the human gut from the colonization of C. Difficile, a diarrhea-causing pathogen that is prone to infecting human patients treated with antibiotics. The host microbes do this by synthesizing inhibitory metabolites from host-derived bile salts[17].
Differentiation of gut microbiota from free-living communities
Because of their ability to transport genes to and from their environment, whole microbial communities are able to adapt at once to new ecological niches[18]. There is perhaps no spatial area with as many distinct niches as the body surface of mammals[19], and this is reflected in comparisons between mammalian microbiota and microbes in free-living communities[2]. Amongst mammals, the microbiota of carnivores, herbivores, and omnivores, are distinct along the lines of these three groups, with herbivores having the highest microbial richness, carnivores having the lowest, and omnivores being in the middle[20]. Microbe communities that are not associated with animal hosts tend to fit into two groups: those in saline conditions and those not in saline conditions[21]. And, despite the ability for horizontal gene transfer to cause rapid evolution in any community, the same microbial phyla tend to perform similar ecological functions in similar environments, no matter how geographically separated they may be[2]. However, vertebrate-associated communities were more than twice as different from each other than the saline and non saline free communities were from themselves. The gut microbiota of mammalian herbivores contributed to the majority of this microbial diversity among vertebrates. Invertebrate microbial communities were also more similar to the saline and non-saline environments than they were to vertebrates, who seem to form a completely distinct environment for microbiomes[2]. Microbes associated with the mammalian gut tend to be fast growing[22], something that may have been important for early relationships between microbes and this new environment. Genera of bacteria that seem to be able to inhabit both free environments and invertebrate guts include Pseudomonadaceae, Enterobacteriales, and Staphylococcaceae[2].
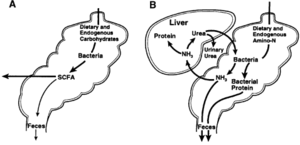
Gut Fermentation
Plants are the most abundant source of food on land[4], but they contain complex carbohydrates like cellulose, hemicellulose and lignin that require more enzymatic power to digest. Herbivores have evolved enlarged fermentation chambers in their guts in order to accomodate microbes to help break down resistant starches, as well as to slow down the passage of food so that there is more time for fermentation to occur. These enlarged chambers can be at the end of the digestive system (hindgut, or post-gastric) like inside elephants, rhinos, gorillas and pigs, or at the start (foregut, or pre-gastric), like inside cows, deer, sheep, and kangaroos[24]. Foregut fermenters house their microbes in a chamber before their stomachs, so the microbes are exposed to food that is still highly nutrient dense. Whilst this means host animals must 'sacrifice' some nutrients to their microbes, it also means that they will eventually end up digesting the bacteria that fall into their stomach, so nothing goes to waste[20]. Ruminants utilize bacteria such as Ruminococcus to digest their food. And in some animals (such as cattle) additional fermentation occurs after the stomach, in the large bowel and cesum, where bacteria produce important vitamins[25].
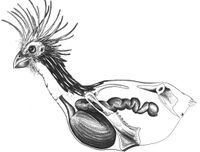
The Hoatzin: a bird with the digestive microbes of a cow
In support for the hypothesis that mammalian gut microbes are largely dependent on diet, one bird that feeds mostly on leaves is a foregut fermenter, and has a similar digestive microbiome to that of cattle[27]. Foregut fermentation occurs mostly in ruminants, such as cows, that have numerous gut chambers and frequently regurgitate and rechew the contents of their digestive system. The hoatzin is the only bird to have evolved a system of foregut fermentation[27][28]. This is an example of convergent evolution between hoatzin and ruminants. The hoatzin is native to South America and, unlike most birds, feeds mostly on young leaves. The bird has an enlarged crop that houses a diverse microbiome, including bacteria, archeaea, and fungi, as well as ciliates, with bacterial phyla including Firmicutes, Bacteroides, Actinobacteria and Proteobacteria, amongst others[29] Filipa Godoy-Vitorino, and her colleagues, compared the bacterial composition of both cow and hoatzin digestive systems in a 2011 study. They found that diet was a larger driver for microbial diversity than phylogenetic lineage. The hoatzin microbiota was more similar to other foregut fermenting animals than the microbiota of chickens and turkeys. However, the cow rumen had higher bacterial richness overall. This could be due to longer retention time of food in the cow rumen, and larger size of the rumen over all[27]. These hypotheses are supported by a study conducted in 2000, showing how the cow rumen was more successful at digesting cellulose than the hoatzin foregut (or crop)[30].
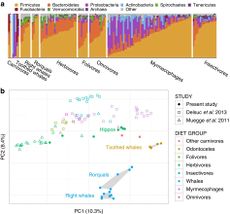
The Residual Foregut in Baleen Whales
In contrast to the hoatzin, baleen whales provide evidence to suggest that phylogeny can play a role in shaping mammalian gut microbiota. Although now carnivorous (preying on fish and crustaceans), whales evolved from terrestrial, deer-like mammals, and still have pre-gastric fermentation chambers[32][33]. Whilst the primary polysaccharide in the diet of a terrestrial herbivore is cellulose, whales must digest the large amounts of chitin found in small bony fish and in the shells of crustaceans[31]. Sanders et al., found that whilst the whale microbiome is similar to that of terrestrial carnivores when it comes to genes related to protein digestion and biosynthesis, with regard to genes involved in carbon and energy metabolism, it has more in common with terrestrial herbivores (based on predicted coding content of shotgun metagenomes in eight baleen whales)[31]. The digestive system of baleen whales has many morphological similarities with those of terrestrial herbivores. As well as a chambered foregut, baleen whales also have a post-gastric fermentation chamber (caecum) that toothed whales do not possess [33]. Although there is little evidence that whales produce methane like terrestrial ruminants, another end-product of microbial fermentation, short-chain fatty acids, have been found in the foreguts of grey and bowhead whales[34]. Sanders et al., also found that the number of genes associated with the metabolism of pyruvate (a major intermediary in fermentation), were far higher in whales than in terrestrial carnivores[31]. Despite the fact that whales share a similar chitin-based diet with large insectivores like anteaters, their gut microbiome is vastly different, as anteaters lack fermentation chambers to house microbes.
Detoxification of Plant Secondary Compounds
Unlike animals, plants cannot run away from other organisms trying to consume them. Whilst moving prey have evolved fast running legs and meticulous camouflage to evade predators, plants must rely on chemical compounds to avoid being eaten. Plant toxins are diverse, and can do anything to consumers from causing mild digestive discomfort, to triggering abortions and death [4]. These compounds are known as plant secondary metabolites (PSMs), and they have a significant impact on the nutritional ecology of mammals that eat them[3].Whilst herbivores can develop enzymes to digest toxins themselves, mammalian genomes are far slower to evolve than those of tiny, fast-replicating bacteria. Microbes are able to digest almost any compound on the planet[35], and since they evolve so quickly and can horizontally transfer genes between species, they are able to occupy any ecological niche within the mammalian gut. This may be one of the reasons that herbivorous mammals have been so successful in many earth ecosystems[4]. However, studies that specifically address the microbiome as a driver of diet diversification in herbivores have been inconclusive[36].
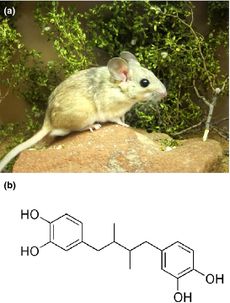
Detoxification of Creosote resin in Desert Woodrats
Study of the relationship between the gut microbiota in The desert woodrat (N. lepida), and the animals tolerance to PSCs on the creosote bush in the mojave desert, is well documented[37][36][5]. The creosote bush (Larrea tridentata) is abundant in the Mojave desert, in the southern United States, because of its resistance to herbivores, drought and ageing [4]. The plant is covered in waxy chemicals that prevent water loss and deter predators. The oldest ring of cloned creosote bushes has been alive for over 11,000 years [38].
Whilst the native population of N. lepida living in the Mojave desert are able to survive on little more than just creosote leaves over the winter and spring months, if a lab rat consumes too much, it dies[39]. Kevin Kohl and his colleagues investigated the differences between woodrat populations in the Mojave desert, where the Creosote bush is present, and the Great Basin Desert, where it is not[5]. Whilst the experienced, creosote eating, Mojave desert population of woodrats tends to express more detoxification enzymes[40], they also have distinct microbial communities in their gut when compared to the woodrats native to the great basin[36]. When Kohl fed the Mojave desert woodrats antibiotics, they were unable to feed on creosote resin and lost >10% of their body mass within 2 weeks, indicating that microbes are largely responsible for the rats' adaptable diet. Conversely, woodrats from the Great Basin population were able to consume only creosote resin whilst maintaining their body weight after receiving a fecal transplant from the experienced rats. Although the specific species responsible for these changes is not known, the antibiotic reduced the abundancy of Spirochaete and Tenericute bacteria[5].
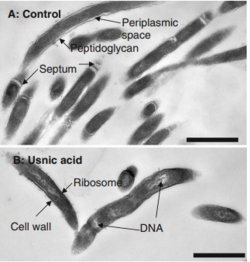
Detoxification of Usnic Acid in Reindeer
Lichens are well known in the microbial world for their iconic symbiosis of fungi and cyanobacteria/green aglae. But they also harbor a poisonous chemical, usnic acid, that provides another example of microbial detoxification in the mammalian gut. This compound functions as an antibiotic for the lichen, as well as an antifeedant (it causes growth retardation and death in cotton leafworms[41]), it also acts as a UV-absorbing pigment to protect the lichen from solar radiation[42]. Usnic acid is toxic to the liver of rats, mice, and humans, and toxic to the skeletal muscles of ruminants, but reindeer are the only animals able to eat and survive on a diet on only lichens[43][44]. One novel strain of bacteria has been identified as a dominant member of the viable reindeer rumen population, Eubacterium rangiferina, a gram-negative, nonmotile rod that was able to actively invade lichen in the lab[44]. Commensal relationships such as these can be immensely valuable, as demonstrated by the deaths of 328 elk in 2004 following the ingestion of usnic acid in the tumbleweed sheild lichen[45]. Elk do not have a lichen-resistant gut microbiome, and could not detoxify the usnic acid in Xanthoparmelia chlorochroa when they were left with no other food source.
Sources of Detoxifying Microbes
Behavioural adaptations of herbivores may help them to alter their gut microbiomes in order to cope with plant toxins. Many mammals, including desert woodrats, are known to engage in geophagia (soil consumption) in order to absorb toxins[46]. This is a possible source of detoxifying bacteria, as soil contains many microbes that are able to degrade phenolic compounds[47]. Woodrats are also known to collect and store feces from larger mammals, such as jackrabbits, cows, and coyote[48], this is another potential source of microbe acquisition. The most likely source of detoxifying microbes, however, is from the plants themselves, where bacteria may have already learned to digest the toxins that herbivorous mammals are trying to eat[49]. 24% of microbes found in the guts of Mojave desert woodrats are also found on the surfaces of creosote leaves[5].
Conclusion
Coevolution is different from other, related processes. It means that two or more different species reciprocate evolutionary adaptations in response to one another, so much so that one organism, eventually, may not be able to survive without the other[50]. It is difficult to know to what degree mammals and their microbiome have developed evolutionary responses to one another throughout time. It is clear that hosts have evolved ways to control their microbiota, and microbes may have evolved in response, but these responses may not be strong enough to provoke a second response from mammalian hosts[7]. Living hosts are different from other environments because they have this ability to evolve, and respond to selection pressures, albeit more slowly than bacteria can. If changes in the gut microbiome lead to reduced host fitness, the host may spread its microbes less than a similar, longer-lived host of greater fitness. These microbes would then have less chance of colonizing other guts[51]. The mammalian gut is a unique microbial habitat because of its high abundance, diversity and flux of different polysaccharides in and out of an anoxic system, and at a constantly controlled temperature. Its ecology is arguably much more complex than any other microbial ecosystem on earth.
See Also
Stomach
Small Intestine
The Hologenome Theory of Evolution
Microbes and Animal Behavior
Intestinal Microflora and Antibiotic Resistance
References
- ↑ Luo, Zhe-Xi. "Transformation and diversification in early mammal evolution." Nature 450.7172 (2007): 1011-1019.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 Ley, Ruth E et al. “Worlds within worlds: evolution of the vertebrate gut microbiota.” Nature reviews. Microbiology vol. 6,10 (2008): 776-88. doi:10.1038/nrmicro1978
- ↑ 3.0 3.1 Dearing, M. Denise, William J. Foley, and Stuart McLean. "The influence of plant secondary metabolites on the nutritional ecology of herbivorous terrestrial vertebrates." Annual review of ecology, evolution, and systematics 36 (2005)
- ↑ 4.0 4.1 4.2 4.3 4.4 Yong, Ed. I contain multitudes: The microbes within us and a grander view of life. Random House, 2016.
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 Kohl, Kevin D., et al. "Gut microbes of mammalian herbivores facilitate intake of plant toxins." Ecology Letters 17.10 (2014): 1238-1246.
- ↑ Kelley, Scott T., and Susanne Dobler. "Comparative analysis of microbial diversity in Longitarsus flea beetles (Coleoptera: Chrysomelidae)." Genetica 139.5 (2011): 541-550.
- ↑ 7.0 7.1 7.2 Foster, Kevin R., et al. "The evolution of the host microbiome as an ecosystem on a leash." Nature 548.7665 (2017): 43-51.
- ↑ 8.0 8.1 Schluter, Jonas, and Kevin R. Foster. "The evolution of mutualism in gut microbiota via host epithelial selection." PLoS biology 10.11 (2012).
- ↑ 9.0 9.1 McLoughlin, Kirstie, et al. "Host selection of microbiota via differential adhesion." Cell host & microbe 19.4 (2016): 550-559.
- ↑ Jarry, Anne, et al. "Subversion of human intestinal mucosa innate immunity by a Crohn’s disease-associated E. coli." Mucosal immunology 8.3 (2015): 572-581.
- ↑ 11.0 11.1 Hooper, Lora V., Dan R. Littman, and Andrew J. Macpherson. "Interactions between the microbiota and the immune system." science 336.6086 (2012): 1268-1273.
- ↑ Caldara, Marina, et al. "Mucin biopolymers prevent bacterial aggregation by retaining cells in the free-swimming state." Current Biology 22.24 (2012): 2325-2330.
- ↑ Rakoff-Nahoum, Seth, et al. "Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis." Cell 118.2 (2004): 229-241.
- ↑ Lamkanfi, Mohamed, and Vishva M. Dixit. "Mechanisms and functions of inflammasomes." Cell 157.5 (2014): 1013-1022.
- ↑ Schluter, Jonas, and Kevin R. Foster. "The evolution of mutualism in gut microbiota via host epithelial selection." PLoS biology 10.11 (2012).
- ↑ Scheuring, István, and Douglas W. Yu. "How to assemble a beneficial microbiome in three easy steps." Ecology letters 15.11 (2012): 1300-1307.
- ↑ Buffie, Charlie G., et al. "Precision microbiome reconstitution restores bile acid mediated resistance to Clostridium difficile." Nature 517.7533 (2015): 205-208.
- ↑ Woese, Carl R. "On the evolution of cells." Proceedings of the National Academy of Sciences 99.13 (2002): 8742-8747.
- ↑ Martiny, Jennifer B. Hughes, et al. "Microbial biogeography: putting microorganisms on the map." Nature Reviews Microbiology 4.2 (2006): 102-112.
- ↑ 20.0 20.1 Ley, Ruth E., et al. "Evolution of mammals and their gut microbes." Science 320.5883 (2008): 1647-1651.
- ↑ Lozupone, Catherine A., and Rob Knight. "Global patterns in bacterial diversity." Proceedings of the National Academy of Sciences 104.27 (2007): 11436-11440.
- ↑ Acinas, Silvia G., et al. "Divergence and redundancy of 16S rRNA sequences in genomes with multiple rrn operons." Journal of bacteriology 186.9 (2004): 2629-2635.
- ↑ Stevens, C. Edward, and Ian D. Hume. "Contributions of microbes in vertebrate gastrointestinal tract to production and conservation of nutrients." Physiological reviews 78.2 (1998): 393-427.
- ↑ Karasov, William H., Carlos Martinez del Rio, and Enrique Caviedes-Vidal. "Ecological physiology of diet and digestive systems." Annual review of physiology 73 (2011): 69-93.
- ↑ Burkholder, Paul R., and Ilda McVeigh. "Synthesis of vitamins by intestinal bacteria." Proceedings of the National Academy of Sciences of the United States of America 28.7 (1942): 285.
- ↑ Grajal, A., and S. D. Strahl. "A bird with the guts to eat leaves." Natural History 8 (1991): 48.
- ↑ 27.0 27.1 27.2 Godoy-Vitorino, Filipa, et al. "Comparative analyses of foregut and hindgut bacterial communities in hoatzins and cows." The ISME journal 6.3 (2012): 531-541.
- ↑ Grajal, Alejandro, et al. "Foregut fermentation in the hoatzin, a neotropical leaf-eating bird." Science 245.4923 (1989): 1236-1238.
- ↑ Godoy-Vitorino, Filipa, et al. "Bacterial community in the crop of the hoatzin, a neotropical folivorous flying bird." Appl. Environ. Microbiol. 74.19 (2008): 5905-5912.
- ↑ Jones, Raymond J., MA Garcia Amado, and M. G. Dominguez-Bello. "Comparison of the digestive ability of crop fluid from the folivorous Hoatzin (Opisthocomus hoazin) and cow rumen fluid with seven tropical forages." Animal feed science and technology 87.3-4 (2000): 287-296.
- ↑ 31.0 31.1 31.2 31.3 Sanders, Jon G., et al. "Baleen whales host a unique gut microbiome with similarities to both carnivores and herbivores." Nature communications 6 (2015): 8285.
- ↑ Gatesy, John, et al. "A phylogenetic blueprint for a modern whale." Molecular phylogenetics and evolution 66.2 (2013): 479-506.
- ↑ 33.0 33.1 Langer, P. "Evidence from the digestive tract on phylogenetic relationships in ungulates and whales." Journal of Zoological Systematics and Evolutionary Research 39.1/2 (2001): 77-90.
- ↑ Herwig, R. P., et al. "Baleen whales: preliminary evidence for forestomach microbial fermentation." Appl. Environ. Microbiol. 47.2 (1984): 421-423.
- ↑ Leahy, Joseph G., and Rita R. Colwell. "Microbial degradation of hydrocarbons in the environment." Microbiology and Molecular Biology Reviews 54.3 (1990): 305-315
- ↑ 36.0 36.1 36.2 Kohl, Kevin D., and M. D. Dearing. "Experience matters: prior exposure to plant toxins enhances diversity of gut microbes in herbivores." Ecology letters 15.9 (2012): 1008-1015.
- ↑ Karasov, William H. "Nutritional bottleneck in a herbivore, the desert wood rat (Neotoma lepida)." Physiological Zoology 62.6 (1989): 1351-1382.
- ↑ Vasek, Frank C. "Creosote bush: Long‐lived clones in the Mojave Desert." American Journal of Botany 67.2 (1980): 246-255.
- ↑ Ríos, Juan Manuel, Antonio Marcelo Mangione, and Jose Carlos Gianello. "Effects of natural phenolic compounds from a desert dominant shrub Larrea divaricata Cav. on toxicity and survival in mice." (2008).
- ↑ Magnanou, E., J. R. Malenke, and M. D. Dearing. "Expression of biotransformation genes in woodrat (Neotoma) herbivores on novel and ancestral diets: identification of candidate genes responsible for dietary shifts." Molecular Ecology 18.11 (2009): 2401-2414.
- ↑ Emmerich, Robert, et al. "Toxicity and antifeedant activity of lichen compounds against the polyphagous herbivorous insect Spodoptera littoralis." Phytochemistry 33.6 (1993): 1389-1394.
- ↑ Bjerke, Jarle Werner, and Tina Dahl. "Distribution patterns of usnic acid-producing lichens along local radiation gradients in West Greenland." Nova Hedwigia 75.3-4 (2002): 487-506.
- ↑ Sundset, Monica A., et al. "Microbial degradation of usnic acid in the reindeer rumen." Naturwissenschaften 97.3 (2010): 273-278.
- ↑ 44.0 44.1 44.2 Sundset, Monica A., et al. "Eubacterium rangiferina, a novel usnic acid-resistant bacterium from the reindeer rumen." Naturwissenschaften 95.8 (2008): 741-749.
- ↑ Roach, John AG, et al. "Determination of usnic acid in lichen toxic to elk by liquid chromatography with ultraviolet and tandem mass spectrometry detection." Journal of agricultural and food chemistry 54.7 (2006): 2484-2490.
- ↑ Krishnamani, Ramanathan, and William C. Mahaney. "Geophagy among primates: adaptive significance and ecological consequences." Animal behaviour 59.5 (2000): 899-915.
- ↑ Blum, Udo, and Steven R. Shafer. "Microbial populations and phenolic acids in soil." Soil Biology and Biochemistry 20.6 (1988): 793-800.
- ↑ Betancourt, Julio L., Thomas R. Van Devender, and Paul Schultz Martin, eds. Packrat middens: the last 40,000 years of biotic change. University of Arizona Press, 1990.
- ↑ Kohl, Kevin D., and M. Denise Dearing. "Wild‐caught rodents retain a majority of their natural gut microbiota upon entrance into captivity." Environmental microbiology reports 6.2 (2014): 191-195.
- ↑ Thompson, John N. Interaction and coevolution. University of Chicago Press, 2014.
- ↑ Ley, Ruth E., Daniel A. Peterson, and Jeffrey I. Gordon. "Ecological and evolutionary forces shaping microbial diversity in the human intestine." Cell 124.4 (2006): 837-848.
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2018, Kenyon College.
